A monoclonal cytolytic T-lymphocyte response observed in a melanoma patient vaccinated with a tumor-specific antigenic peptide encoded by gene MAGE-3 - PubMed (original) (raw)
Case Reports
. 2001 Aug 28;98(18):10290-5.
doi: 10.1073/pnas.161260098. Epub 2001 Aug 21.
Affiliations
- PMID: 11517302
- PMCID: PMC56954
- DOI: 10.1073/pnas.161260098
Case Reports
A monoclonal cytolytic T-lymphocyte response observed in a melanoma patient vaccinated with a tumor-specific antigenic peptide encoded by gene MAGE-3
P G Coulie et al. Proc Natl Acad Sci U S A. 2001.
Abstract
Vaccination of melanoma patients with tumor-specific antigens recognized by cytolytic T lymphocytes (CTL) produces significant tumor regressions in a minority of patients. These regressions appear to occur in the absence of massive CTL responses. To detect low-level responses, we resorted to antigenic stimulation of blood lymphocyte cultures in limiting dilution conditions, followed by tetramer analysis, cloning of the tetramer-positive cells, and T-cell receptor (TCR) sequence analysis of the CTL clones that showed strict specificity for the tumor antigen. A monoclonal CTL response against a MAGE-3 antigen was observed in a melanoma patient, who showed partial rejection of a large metastasis after treatment with a vaccine containing only the tumor-specific antigenic peptide. Tetramer analysis after in vitro restimulation indicated that about 1/40,000 postimmunization CD8(+) blood lymphocytes were directed against the antigen. The same TCR was present in all of the positive microcultures. TCR evaluation carried out directly on blood lymphocytes by PCR amplification led to a similar frequency estimate after immunization, whereas the TCR was not found among 2.5 x 10(6) CD8(+) lymphocytes collected before immunization. Our results prove unambiguously that vaccines containing only a tumor-specific antigenic peptide can elicit a CTL response. Even though they provide no information about the effector mechanisms responsible for the observed reduction in tumor mass in this patient, they would suggest that low-level CTL responses can initiate tumor rejection.
Figures
Figure 1
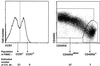
Detection of anti-MAGE-3.A1 CTLp. (A) Clinical evolution of patient CP64. (B) Detection of anti-MAGE-3.A1 CTL in postimmunization PBMC. PBMC were thawed, incubated with peptide MAGE-3.A1, washed, and distributed at 3.106 cells/well in the presence of IL-2, -4, and -7. On day 7, the cultures were restimulated with peptide and cytokines. On days 7 and 13, the cells were incubated with the A1/MAGE-3 tetramer coupled to PE and with a control HLA-A1 tetramer containing an influenza peptide and coupled to APC. Anti-CD3 antibodies coupled to FITC and anti-CD8 antibodies coupled to peridinin chlorophyll protein were then added. Cells were washed, fixed, and analyzed by flow cytometry. One million events were acquired, but the plots include only the CD3+CD8+ lymphocytes. The lytic activity of CTL clones, derived from two populations of cells labeled with tetramer, was tested on HLA-A1 EBV-B cells incubated or not with the MAGE-3.A1 peptide (2 μM), natural killer target cells K562, and allogenic melanoma cells MZ2-MEL, which naturally express the MAGE-3.A1 antigen. The pattern of lysis of two representative clones is shown. (C) Estimation of the frequency of anti-MAGE-3.A1 CTLp in postimmunization PBMC. Thirty-six cultures were set up with 160,000 Post II PBMC stimulated with the MAGE-3.A1 peptide and IL-2, -4, and -7. The lymphocytes were restimulated on day 7 by the addition of peptide and cytokines and labeled on day 15 with tetramers and anti-CD3 and anti-CD8 antibodies. Only CD3+CD8+ lymphocytes are included in the plots. Clusters of lymphocytes specifically labeled with the A1/MAGE-3 tetramer are boxed, and their proportion among the CD3+CD8+ cells is indicated. (D) Analysis of prevaccination PBMC. Five cultures of 3 million PBMC were set up and analyzed as in C.
Figure 2
Phenotype of PBMC expressing TCR 48. Postimmunization (Post II) PBMC were thawed and labeled either with anti-CD45RO antibodies coupled to FITC and anti-CD45RA antibodies coupled to PE, or with an anti-CCR7 IgM antibody followed by biotinylated anti-IgM antibodies and streptavidin coupled to PE. The cells were immediately sorted by flow cytometry. Sorted populations were divided in groups and cDNA obtained from each group was tested for the TCRα and β clonotypic PCR. Numbers of CTL 48 were calculated with the Poisson distribution.
Similar articles
- Polyclonal CTL responses observed in melanoma patients vaccinated with dendritic cells pulsed with a MAGE-3.A1 peptide.
Godelaine D, Carrasco J, Lucas S, Karanikas V, Schuler-Thurner B, Coulie PG, Schuler G, Boon T, Van Pel A. Godelaine D, et al. J Immunol. 2003 Nov 1;171(9):4893-7. doi: 10.4049/jimmunol.171.9.4893. J Immunol. 2003. PMID: 14568970 - Monoclonal anti-MAGE-3 CTL responses in melanoma patients displaying tumor regression after vaccination with a recombinant canarypox virus.
Karanikas V, Lurquin C, Colau D, van Baren N, De Smet C, Lethé B, Connerotte T, Corbière V, Demoitié MA, Liénard D, Dréno B, Velu T, Boon T, Coulie PG. Karanikas V, et al. J Immunol. 2003 Nov 1;171(9):4898-904. doi: 10.4049/jimmunol.171.9.4898. J Immunol. 2003. PMID: 14568971 - Antigen spreading contributes to MAGE vaccination-induced regression of melanoma metastases.
Corbière V, Chapiro J, Stroobant V, Ma W, Lurquin C, Lethé B, van Baren N, Van den Eynde BJ, Boon T, Coulie PG. Corbière V, et al. Cancer Res. 2011 Feb 15;71(4):1253-62. doi: 10.1158/0008-5472.CAN-10-2693. Epub 2011 Jan 7. Cancer Res. 2011. PMID: 21216894 - Cytolytic T-cell responses of cancer patients vaccinated with a MAGE antigen.
Coulie PG, Karanikas V, Lurquin C, Colau D, Connerotte T, Hanagiri T, Van Pel A, Lucas S, Godelaine D, Lonchay C, Marchand M, Van Baren N, Boon T. Coulie PG, et al. Immunol Rev. 2002 Oct;188:33-42. doi: 10.1034/j.1600-065x.2002.18804.x. Immunol Rev. 2002. PMID: 12445279 Review. - Antitumor cytotoxic T-lymphocyte response in human lung carcinoma: identification of a tumor-associated antigen.
Mami-Chouaib F, Echchakir H, Dorothée G, Vergnon I, Chouaib S. Mami-Chouaib F, et al. Immunol Rev. 2002 Oct;188:114-21. doi: 10.1034/j.1600-065x.2002.18810.x. Immunol Rev. 2002. PMID: 12445285 Review.
Cited by
- Diversity and recognition efficiency of T cell responses to cancer.
Stuge TB, Holmes SP, Saharan S, Tuettenberg A, Roederer M, Weber JS, Lee PP. Stuge TB, et al. PLoS Med. 2004 Nov;1(2):e28. doi: 10.1371/journal.pmed.0010028. Epub 2004 Nov 30. PLoS Med. 2004. PMID: 15578105 Free PMC article. Clinical Trial. - Immunotherapy of distant metastatic disease.
Schadendorf D, Algarra SM, Bastholt L, Cinat G, Dreno B, Eggermont AM, Espinosa E, Guo J, Hauschild A, Petrella T, Schachter J, Hersey P. Schadendorf D, et al. Ann Oncol. 2009 Aug;20 Suppl 6(Suppl 6):vi41-50. doi: 10.1093/annonc/mdp253. Ann Oncol. 2009. PMID: 19617297 Free PMC article. Review. - Analysis of a rare melanoma patient with a spontaneous CTL response to a MAGE-A3 peptide presented by HLA-A1.
Hanagiri T, van Baren N, Neyns B, Boon T, Coulie PG. Hanagiri T, et al. Cancer Immunol Immunother. 2006 Feb;55(2):178-84. doi: 10.1007/s00262-005-0063-0. Epub 2005 Sep 27. Cancer Immunol Immunother. 2006. PMID: 16187089 Free PMC article. - Biological response modifiers in cancer.
Reang P, Gupta M, Kohli K. Reang P, et al. MedGenMed. 2006 Nov 14;8(4):33. MedGenMed. 2006. PMID: 17415315 Free PMC article. Review. - A new dendritic cell vaccine generated with interleukin-3 and interferon-beta induces CD8+ T cell responses against NA17-A2 tumor peptide in melanoma patients.
Trakatelli M, Toungouz M, Blocklet D, Dodoo Y, Gordower L, Laporte M, Vereecken P, Sales F, Mortier L, Mazouz N, Lambermont M, Goldman S, Coulie P, Goldman M, Velu T. Trakatelli M, et al. Cancer Immunol Immunother. 2006 Apr;55(4):469-74. doi: 10.1007/s00262-005-0056-z. Epub 2005 Aug 20. Cancer Immunol Immunother. 2006. PMID: 16133111 Free PMC article. Clinical Trial.
References
- van der Bruggen P, Traversari C, Chomez P, Lurquin C, De Plaen E, Van den Eynde B, Knuth A, Boon T. Science. 1991;254:1643–1647. - PubMed
- Van den Eynde B, van der Bruggen P. Curr Opin Immunol. 1997;9:684–693. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials