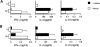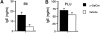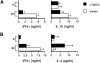Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis - PubMed (original) (raw)
Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis
A K Singh et al. J Exp Med. 2001.
Abstract
Experimental autoimmune encephalomyelitis (EAE) serves as a prototypic model for T cell-mediated autoimmunity. V(alpha)14 natural killer T (NKT) cells are a subset of T lymphocytes that recognize glycolipid antigens presented by the nonpolymorphic major histocompatibility complex (MHC) class I-like protein CD1d. Here, we show that activation of V(alpha)14 NKT cells by the glycosphingolipid alpha-galactosylceramide (alpha-GalCer) protects susceptible mice against EAE. beta-GalCer, which binds CD1d but is not recognized by NKT cells, failed to protect mice against EAE. Furthermore, alpha-GalCer was unable to protect CD1d knockout (KO) mice against EAE, indicating the requirement for an intact CD1d antigen presentation pathway. Protection of disease conferred by alpha-GalCer correlated with its ability to suppress myelin antigen-specific Th1 responses and/or to promote myelin antigen-specific Th2 cell responses. alpha-GalCer was unable to protect IL-4 KO and IL-10 KO mice against EAE, indicating a critical role for both of these cytokines. Because recognition of alpha-GalCer by NKT cells is phylogenetically conserved, our findings have identified NKT cells as novel target cells for treatment of inflammatory diseases of the central nervous system.
Figures
Figure 1.
Analysis of Vα14 NKT cells in EAE-susceptible mice. (A) Numbers of CD1d/α-GalCer tetramer-positive cells in EAE-susceptible mice. Liver mononuclear cells from the indicated mice were stained with APC-labeled CD1d/α-GalCer tetramers and FITC-labeled anti-TCR-β antibodies and analyzed by flow cytometry. Numbers indicate the percentage of TCR-β+ tetramer+ cells. Representative data are shown from three separate experiments. (B) In vitro responses of spleen cells from EAE-susceptible mice to α-GalCer. Splenocytes (2 × 105) from the indicated mice were cultured with 50 ng/ml of α-GalCer. 3 d later proliferation was measured by [3H]thymidine incorporation and cytokine production in the culture supernatant was measured by ELISA. Data represent the mean ± SE of three mice per group. Representative data are shown from three separate experiments.
Figure 2.
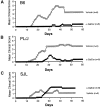
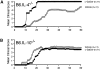
α-GalCer protects susceptible mice against EAE. EAE was induced in B6 mice (A) by immunization with MOG35–55 peptide, and in PL/J (B) and SJL mice (C) by immunization with MBP, as described in Materials and Methods. Mice were treated with α-GalCer (6 μg/injection) or vehicle (0.025% polysorbate-20 in PBS) on days 0, 4, and 7 after immunization. Representative data from several independent experiments are shown.
Figure 3.
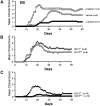
The capacity of α-GalCer to protect mice against EAE is mediated by activation of CD1d-restricted NKT cells. (A) β-GalCer, which binds with CD1d but does not activate NKT cells, fails to protect mice against EAE. B6 mice were immunized with MOG35–55 peptide and treated with α-GalCer (6 μg/injection), β-GalCer (6 μg/injection), or vehicle. (B) CD1d deficiency does not exacerbate EAE in B6 mice. B6.CD1d +/+ and B6.CD1d −/− mice from the 10th backcross to B6 were immunized with MOG35–55 peptide. (C) Protection against EAE requires CD1d expression in mice. EAE was induced in B6.CD1d +/+ and B6.CD1d −/− mice by immunization with MOG35–55. Mice were treated with α-GalCer (4 μg/injection) on days 0 and 7.
Figure 4.
α-GalCer promotes antigen-specific Th2 responses in mice immunized with CNS antigens. B6 mice (A) were immunized with MOG35–55 and PL/J mice (B) were immunized with MBP to induce EAE. Mice were treated with α-GalCer or vehicle as described in the legend to Fig. 2. At day 11 after immunization cells from inguinal and axillary lymph nodes were cultured with the indicated concentrations (in μg/ml) of MOG35–55 peptide (A) or MBP (B). After 60 h of culture, supernatants were collected and cytokine levels were measured by ELISA. Data shown represent the mean values ± SE of 6 mice in each experimental group. Representative data are shown from three separate experiments.
Figure 5.
IgE antibody production in the serum of mice immunized with CNS antigens and treated with α-GalCer. EAE was induced in B6 mice (A) by immunization with MOG35–55 peptide and in PL/J (B) mice by immunization with MBP. Mice were treated with α-GalCer or vehicle as described in the legend to Fig. 2. Mice were bled on day 11 after immunization and total IgE levels in the serum were measured by ELISA. Data shown represent the mean values ± SE of four (A) or six (B) mice in each experimental group. Representative data are shown from two separate experiments.
Figure 6.
α-GalCer fails to protect IL-4 KO and IL-10 KO mice against EAE. B6.IL-4 −/− (A) and B6.IL-10 −/− (B) mice were immunized with MOG35–55 peptide to induce EAE and treated with α-GalCer or vehicle as described in the legend to Fig. 2. In parallel experiments α-GalCer effectively protected wild-type B6 mice against EAE (data not shown). Representative data are shown from three separate experiments.
Figure 7.
CNS antigen-specific responses in IL-4 KO and IL-10 KO mice immunized with MOG35–55 peptides and treated with α-GalCer. B6.IL-4 −/− (A) and B6.IL-10 −/− (B) mice were immunized with MOG35–55 peptide to induce EAE and treated with α-GalCer or vehicle as described in the legend to Fig. 2. At day 11 after immunization cells from inguinal and axillary lymph nodes were cultured with the indicated concentrations (in μg/ml) of MOG35–55 peptide (A) or MBP (B). After 60 h of culture, supernatants were collected and cytokine levels were measured by ELISA. Data shown represent the mean values ± SE of four mice in each experimental group. Representative data are shown from three separate experiments.
Similar articles
- Activation of natural killer T cells potentiates or prevents experimental autoimmune encephalomyelitis.
Jahng AW, Maricic I, Pedersen B, Burdin N, Naidenko O, Kronenberg M, Koezuka Y, Kumar V. Jahng AW, et al. J Exp Med. 2001 Dec 17;194(12):1789-99. doi: 10.1084/jem.194.12.1789. J Exp Med. 2001. PMID: 11748280 Free PMC article. - A subset of NKT cells that lacks the NK1.1 marker, expresses CD1d molecules, and autopresents the alpha-galactosylceramide antigen.
Hameg A, Apostolou I, Leite-De-Moraes M, Gombert JM, Garcia C, Koezuka Y, Bach JF, Herbelin A. Hameg A, et al. J Immunol. 2000 Nov 1;165(9):4917-26. doi: 10.4049/jimmunol.165.9.4917. J Immunol. 2000. PMID: 11046017 - Regulation of immune responses by CD1d-restricted natural killer T cells.
Van Kaer L. Van Kaer L. Immunol Res. 2004;30(2):139-53. doi: 10.1385/IR:30:2:139. Immunol Res. 2004. PMID: 15477656 Review. - Quantitative and qualitative differences in the in vivo response of NKT cells to distinct alpha- and beta-anomeric glycolipids.
Parekh VV, Singh AK, Wilson MT, Olivares-Villagómez D, Bezbradica JS, Inazawa H, Ehara H, Sakai T, Serizawa I, Wu L, Wang CR, Joyce S, Van Kaer L. Parekh VV, et al. J Immunol. 2004 Sep 15;173(6):3693-706. doi: 10.4049/jimmunol.173.6.3693. J Immunol. 2004. PMID: 15356115 - Application of natural killer T cells in antitumor immunotherapy.
Hong C, Park SH. Hong C, et al. Crit Rev Immunol. 2007;27(6):511-25. doi: 10.1615/critrevimmunol.v27.i6.20. Crit Rev Immunol. 2007. PMID: 18197797 Review.
Cited by
- NKT cell stimulation with α-galactosylceramide results in a block of Th17 differentiation after intranasal immunization in mice.
Zygmunt BM, Weissmann SF, Guzman CA. Zygmunt BM, et al. PLoS One. 2012;7(1):e30382. doi: 10.1371/journal.pone.0030382. Epub 2012 Jan 26. PLoS One. 2012. PMID: 22291945 Free PMC article. - Mouse V alpha 14i natural killer T cells are resistant to cytokine polarization in vivo.
Matsuda JL, Gapin L, Baron JL, Sidobre S, Stetson DB, Mohrs M, Locksley RM, Kronenberg M. Matsuda JL, et al. Proc Natl Acad Sci U S A. 2003 Jul 8;100(14):8395-400. doi: 10.1073/pnas.1332805100. Epub 2003 Jun 26. Proc Natl Acad Sci U S A. 2003. PMID: 12829795 Free PMC article. - Overview of a HLA-Ig based "Lego-like system" for T cell monitoring, modulation and expansion.
Oelke M, Schneck JP. Oelke M, et al. Immunol Res. 2010 Jul;47(1-3):248-56. doi: 10.1007/s12026-009-8156-z. Immunol Res. 2010. PMID: 20087680 Free PMC article. Review. - Adjuvants of immunity: harnessing innate immunity to promote adaptive immunity.
Bendelac A, Medzhitov R. Bendelac A, et al. J Exp Med. 2002 Mar 4;195(5):F19-23. doi: 10.1084/jem.20020073. J Exp Med. 2002. PMID: 11877490 Free PMC article. No abstract available. - Islet allograft tolerance in the absence of invariant natural killer T cells.
Chu X, Kilpatrick E, Xiao X, Liu W, Demirci G, Exley M, Li XC. Chu X, et al. Clin Immunol. 2011 Dec;141(3):268-72. doi: 10.1016/j.clim.2011.09.003. Epub 2011 Sep 23. Clin Immunol. 2011. PMID: 21996456 Free PMC article.
References
- Steinman, L. 1999. Assessment of animal models for MS and demyelinating disease in the design of rational therapy. Neuron. 24:511–514. - PubMed
- Powell, M.B., D. Mitchell, J. Lederman, J. Buckmeier, S.S. Zamvil, M. Graham, N.H. Ruddle, and L. Steinman. 1990. Lymphotoxin and tumor necrosis factor-α production by myelin basic protein-specific T cell clones correlates with encephalitogenicity. Int. Immunol. 2:539–544. - PubMed
- Begolka, W.S., C.L. Vanderlugt, S.M. Rahbe, and S.D. Miller. 1998. Differential expression of inflammatory cytokines parallels progression of central nervous system pathology in two clinically distinct models of multiple sclerosis. J. Immunol. 161:4437–4446. - PubMed
- Issazadeh, S., A. Ljungdahl, B. Hojeberg, M. Mustafa, and T. Olsson. 1995. Cytokine production in the central nervous system of Lewis rats with experimental autoimmune encephalomyelitis: dynamics of mRNA expression for interleukin-10, interleukin-12, cytolysin, tumor necrosis factor alpha and tumor necrosis factor beta. J. Neuroimmunol. 61:205–212. - PubMed
- Issazadeh, S., V. Navikas, M. Schaub, M. Sayegh, and S. Khoury. 1998. Kinetics of expression of costimulatory molecules and their ligands in murine relapsing experimental autoimmune encephalomyelitis in vivo. J. Immunol. 161:1104–1112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials