Distinct roles of VEGFR-1 and VEGFR-2 in the aberrant hematopoiesis associated with elevated levels of VEGF - PubMed (original) (raw)
Distinct roles of VEGFR-1 and VEGFR-2 in the aberrant hematopoiesis associated with elevated levels of VEGF
Yuhui Huang et al. Blood. 2007.
Abstract
Vascular endothelial growth factor (VEGF), a major factor in tumor-host interactions, plays a critical role in the aberrant hematopoiesis observed in cancer-bearing hosts. To dissect the roles of VEGF receptor (VEGFR)-1 and VEGFR-2 in cancer-associated hematopoiesis in vivo, we selectively stimulated VEGFR-1 and VEGFR-2 by continuous infusion of receptor-specific ligands or selective blockade with VEGF receptor-specific antibodies in mice infused with recombinant VEGF at levels observed in tumor-bearing animals. We found that the effect of VEGF on the accumulation of Gr1(+)CD11b(+) cells is mediated by VEGFR-2, but that the 2 receptors have opposite effects on lymphocyte development. Pathophysiologic levels of VEGF strongly inhibit T-cell development via VEGFR-2, whereas VEGFR-1 signaling decreases this inhibition. VEGFR-1, and not VEGFR-2, signaling is responsible for the observed increase of splenic B cells. Both receptors are capable of inhibiting dendritic cell function. These data suggest that most of observed aberrant hematopoiesis caused by excess tumor-derived VEGF is mediated by VEGFR-2, and VEGFR-1 alone has very limited independent effects but clearly both positively and negatively modulates the effects of VEGFR-2. Our findings suggest that selective blockade of VEGFR-2 rather than of both receptors may optimally overcome the adverse hematologic consequences of elevated VEGF levels found in malignancy.
Figures
Figure 1
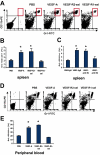
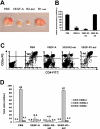
Immature myeloid cells (Gr1+CD11b+) were increased in spleen and peripheral blood of VEGF-pump mice. The 8- to 10-week-old Balb/c mice were given a continuous infusion of PBS, rhVEGF-A (50 ng/hr), VEGFR2-sel (KDR-sel, 50 ng/hr), or VEGFR1-sel (Flt-sel, 50 ng/hr) over a period of 28 days. (A) Splenocytes were analyzed by FACS for the expression of Gr1 and CD11b. The representative FACS plots were shown. The first plot is the isotype control. (B) The percentage of Gr1+CD11b+ cells in spleens. (C) The 8- to 10-week-old Balb/c mice given a continuous infusion of PBS treated with rat IgG or infusions of rhVEGF-A (50 ng/hr) were treated with rat IgG, anti-R1 (MF1), or anti-R2 (DC101), 800 μg every 3 days over a period of 28 days. The percentage of Gr1+CD11b+ cells in spleens is shown. The data (mean ± standard error of the mean [SEM]; n ≥ 5) are repeated 2 times. (D) Red blood cells from murine peripheral blood were lysed and then Gr1+CD11b+ cell population was analyzed by FACS. The representative FACS plots are shown. The first plot is the isotype control. (E) The percentage of Gr1+CD11b+ cells in peripheral blood. All of the other data (mean ± SEM, n ≥ 3) repeat 3 times. *P < .05; **P < .01.
Figure 2
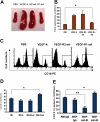
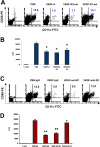
VEGF increases splenic B cells via VEGFR-1 and VEGFR-2 has opposite effect. The 8- to 10-week-old Balb/c mice were given PBS or VEGF receptor agonists for 28 days as described in Figure 1. (A) The representative spleens from PBS, VEGF-A, VEGFR2-sel, and VEGFR1-sel mice (from left to right). (B) The total splenocytes of spleens from the pump mice. (C) The splenocytes were analyzed for expression of CD19. The representative FACS histograms were shown. (D) Total number of B cells (CD19+) in spleens. (E) Balb/c mice were given PBS treated with rat IgG, or rhVEGF-treated rat IgG, anti-R1, or anti-R2 as described in Figure 1C. Total number of B cells (CD19+) in spleens is shown. The data (mean ± SEM, n ≥ 5) repeat 2 times. All of the other data (mean ± SEM, n ≥ 3) repeat 3 times. *P < .05; **P < .01.
Figure 3
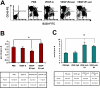
VEGF promotes bone marrow B-cell development via VEGFR-1 and VEGFR-2 has the opposite effect. (A) Flow cytometric analysis of bone marrow cells expressed CD43 and B220. The representative FACS plots were shown. The first plot is the isotype control. (B) The proportion of pre-B/pro-B (left) and the percentage of B-cell subsets (right) in total bone marrow cells: pro-B cells (CD43+B220+) and pre-B cells (CD43−B220+). The data (mean ± SEM, n ≥ 3) repeat 3 times. (C) Balb/c mice were given PBS treated with rat IgG, or rhVEGF-treated rat IgG, anti-R1 or anti-R2 as described in Figure 1C. The proportion of pre-B/pro-B (left) and the percentage of B-cell subtypes (right) in total bone marrow are shown. The data (mean ± SEM, n ≥ 5) repeat 2 times. *P < .05; **P < .01.
Figure 4
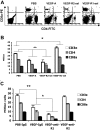
VEGF decreases splenic T cells via VEGFR-2 and VEGFR-1 slightly decreases the inhibition of VEGFR-2. Age-matched Balb/c mice were given PBS or VEGF receptor agonists for 28 days as described in Figure 1. (A) Flow cytometric analysis of the expression of CD4 and CD8a in splenocytes. The representative FACS plots were shown. The first plot is the isotype control. (B) The total numbers of T-cell subsets in spleens. The data (mean ± SEM, n ≥ 3) repeat 3 times. (C) Balb/c mice were given PBS treated with rat IgG, or rhVEGF-treated rat IgG, anti-R1, or anti-R2 as described in Figure 1C. The total numbers of T-cell subsets in spleen were shown. The data (mean ± SEM, n ≥ 5) repeat 2 times. *P < .05; **P < .01.
Figure 5
VEGF inhibits thymic T-cell development via VEGFR-2. (A) The representative thymuses from PBS, VEGF-A, VEGFR2-sel, and VEGFR1-sel mice (from left to right). (B) The total thymocytes. (C) Flow cytometric analysis of thymocytes expressed CD4 and CD8a in CD3elow/− cells. The representative FACS plots were shown. The first plot is the isotype control. (D) The total numbers of T-cell subtypes in thymus. All of the data (mean ± SEM, n ≥ 3) repeat 3 times. *P < .05; **P < .01.
Figure 6
VEGF inhibits the ability of DCs to stimulate allogeneic T-cell proliferation via both of VEGFR-1 and VEGFR-2, and blockade of VEGFR-2 is sufficient to restore DC function. The 8- to 10-week-old Balb/c mice were given PBS or VEGF receptor agonists for 28 days as described in Figure 1. (A) Expression of CD11c and CD86 in CD3e−B220− splenocytes was analyzed by FACS. The representative FACS plots were shown. The numbers represent the means of the percentage of indicated cell fractions. The first plot is the isotype control. (B) The ability of DCs to stimulate allogeneic T-cell proliferation. DCs were isolated from splenocytes with CD11c Microbeads. DCs (105) were incubated with 4 × 105 T cells (C57BL/6) for 4 days and 3H-thymidine uptake was analyzed in triplicate. (C) Balb/c mice were given PBS treated with rat IgG, or rhVEGF-treated rat IgG, anti-R1, or anti-R2 as described in Figure 1C. Expression of CD11c and CD86 in CD3e−B220− splenocytes was analyzed by FACS. The representative FACS plots were shown. The numbers represent the means of the percentage of indicated cell fractions. The first plot is the isotype control. (D) The ability of DCs to stimulate allogeneic T-cell proliferation. DCs were isolated from splenocytes with CD11c Microbeads. DCs (105) were incubated with 4 × 105 T cells (C57BL/6) for 4 days and 3H-thymidine uptake was analyzed in triplicate. The data (mean ± SEM, n = 3) repeat 2 times.
Figure 7
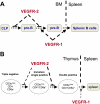
A model for the opposite effects of VEGFR-1 and VEGFR-2 on lymphoid cell development. (A) VEGF suppresses the progression from common lymphoid progenitors (CLP) to pro-B cells and from pro-B to pre-B cells via VEGFR-2; whereas VEGF promotes the progression from pro-B to pre-B cells, and the exit from bone marrow to spleen via VEGFR-1. (B) VEGF suppresses the progression from triple-negative to double-positive, especially from intermediate single-positive to double-positive thymocytes via VEGFR-2, and VEGF promotes the mobilization of related cells via VEGFR-1 (such as the exit of the mature single positive T cells from thymus to spleen).
Similar articles
- A novel snake venom vascular endothelial growth factor (VEGF) predominantly induces vascular permeability through preferential signaling via VEGF receptor-1.
Takahashi H, Hattori S, Iwamatsu A, Takizawa H, Shibuya M. Takahashi H, et al. J Biol Chem. 2004 Oct 29;279(44):46304-14. doi: 10.1074/jbc.M403687200. Epub 2004 Aug 24. J Biol Chem. 2004. PMID: 15328352 - Vascular endothelial growth factor receptor-1 regulates postnatal angiogenesis through inhibition of the excessive activation of Akt.
Nishi J, Minamino T, Miyauchi H, Nojima A, Tateno K, Okada S, Orimo M, Moriya J, Fong GH, Sunagawa K, Shibuya M, Komuro I. Nishi J, et al. Circ Res. 2008 Aug 1;103(3):261-8. doi: 10.1161/CIRCRESAHA.108.174128. Epub 2008 Jun 26. Circ Res. 2008. PMID: 18583712 - Vascular endothelial growth factor receptor-1 and receptor-2 initiate a phosphatidylinositide 3-kinase-dependent clonogenic response in acute myeloid leukemia cells.
List AF, Glinsmann-Gibson B, Stadheim C, Meuillet EJ, Bellamy W, Powis G. List AF, et al. Exp Hematol. 2004 Jun;32(6):526-35. doi: 10.1016/j.exphem.2004.03.005. Exp Hematol. 2004. PMID: 15183893 - The role of vascular endothelial growth factor (VEGF) in tumor angiogenesis and early clinical development of VEGF-receptor kinase inhibitors.
Verheul HM, Pinedo HM. Verheul HM, et al. Clin Breast Cancer. 2000 Sep;1 Suppl 1:S80-4. doi: 10.3816/cbc.2000.s.015. Clin Breast Cancer. 2000. PMID: 11970755 Review.
Cited by
- Anti-angiogenic Agents in Combination With Immune Checkpoint Inhibitors: A Promising Strategy for Cancer Treatment.
Song Y, Fu Y, Xie Q, Zhu B, Wang J, Zhang B. Song Y, et al. Front Immunol. 2020 Aug 25;11:1956. doi: 10.3389/fimmu.2020.01956. eCollection 2020. Front Immunol. 2020. PMID: 32983126 Free PMC article. Review. - CD4+VEGFR1(HIGH) T cell as a novel Treg subset regulates inflammatory bowel disease in lymphopenic mice.
Shin JY, Yoon IH, Lim JH, Shin JS, Nam HY, Kim YH, Cho HS, Hong SH, Kim JS, Lee WW, Park CG. Shin JY, et al. Cell Mol Immunol. 2015 Sep;12(5):592-603. doi: 10.1038/cmi.2015.71. Epub 2015 Jul 27. Cell Mol Immunol. 2015. PMID: 26211666 Free PMC article. - The immunosuppressive factors IL-10, TGF-β, and VEGF do not affect the antigen-presenting function of CD40-activated B cells.
Shimabukuro-Vornhagen A, Draube A, Liebig TM, Rothe A, Kochanek M, von Bergwelt-Baildon MS. Shimabukuro-Vornhagen A, et al. J Exp Clin Cancer Res. 2012 May 16;31(1):47. doi: 10.1186/1756-9966-31-47. J Exp Clin Cancer Res. 2012. PMID: 22592077 Free PMC article. - Hot and Cold Tumors: Is Endoglin (CD105) a Potential Target for Vessel Normalization?
Ollauri-Ibáñez C, Ayuso-Íñigo B, Pericacho M. Ollauri-Ibáñez C, et al. Cancers (Basel). 2021 Mar 28;13(7):1552. doi: 10.3390/cancers13071552. Cancers (Basel). 2021. PMID: 33800564 Free PMC article. Review. - Cancer-associated myeloproliferation: old association, new therapeutic target.
Wilcox RA. Wilcox RA. Mayo Clin Proc. 2010 Jul;85(7):656-63. doi: 10.4065/mcp.2010.0077. Mayo Clin Proc. 2010. PMID: 20592171 Free PMC article. Review.
References
- Shalaby F, Rossant J, Yamaguchi TP, et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature. 1995;376:62–66. - PubMed
- Fong GH, Rossant J, Gertsenstein M, Breitman ML. Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature. 1995;376:66–70. - PubMed
- Ferrara N, Carver-Moore K, Chen H, et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature. 1996;380:439–442. - PubMed
- Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–676. - PubMed
- Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol. 2005;23:1011–1027. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials