Endothelial cell lumen and vascular guidance tunnel formation requires MT1-MMP-dependent proteolysis in 3-dimensional collagen matrices - PubMed (original) (raw)
Endothelial cell lumen and vascular guidance tunnel formation requires MT1-MMP-dependent proteolysis in 3-dimensional collagen matrices
Amber N Stratman et al. Blood. 2009.
Abstract
Here we show that endothelial cells (EC) require matrix type 1-metalloproteinase (MT1-MMP) for the formation of lumens and tube networks in 3-dimensional (3D) collagen matrices. A fundamental consequence of EC lumen formation is the generation of vascular guidance tunnels within collagen matrices through an MT1-MMP-dependent proteolytic process. Vascular guidance tunnels represent a conduit for EC motility within these spaces (a newly remodeled 2D matrix surface) to both assemble and remodel tube structures. Interestingly, it appears that twice as many tunnel spaces are created than are occupied by tube networks after several days of culture. After tunnel formation, these spaces represent a 2D migratory surface within 3D collagen matrices allowing for EC migration in an MMP-independent fashion. Blockade of EC lumenogenesis using inhibitors that interfere with the process (eg, integrin, MMP, PKC, Src) completely abrogates the formation of vascular guidance tunnels. Thus, the MT1-MMP-dependent proteolytic process that creates tunnel spaces is directly and functionally coupled to the signaling mechanisms required for EC lumen and tube network formation. In summary, a fundamental and previously unrecognized purpose of EC tube morphogenesis is to create networks of matrix conduits that are necessary for EC migration and tube remodeling events critical to blood vessel assembly.
Figures
Figure 1
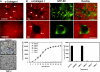
MMP activity is required for EC lumen formation and generation of vascular guidance tunnels in 3D collagen matrices. (A) EC cultures were established with or without the addition of the proteinase inhibitors GM6001 (5 μM; top panels) or recombinant TIMP-3 (5 μg/mL; bottom panels). Collagen gels were fixed at 24 hours and processed for immunostaining of the collagen type I matrix (top panels) or were fixed after 48 hours (bottom panels). (Top panels) White arrows indicate the outline of vascular guidance tunnels. Bar equals 50 μm. (Bottom panels) Representative light microscopy images are shown demonstrating quantification of EC lumen formation with and without TIMP-3 addition. Bar equals 100 μm. (B) GFP-ECs were seeded within collagen matrices and allowed to form lumens and tube networks. Cultures were fixed at 96 hours and immunostained for the collagen type I matrix using a collagen type I monoclonal antibody and an Alexa Fluor 594 conjugated secondary antibody. Representative fluorescent images are shown which illustrate that ECs undergo morphogenesis within vascular guidance tunnels. Arrows denote the borders of vascular guidance tunnels. 10×, bar equals 100 μm; 40×, bar equals 25 μm. (C) ECs were suspended in 3D collagen gels and allowed to undergo morphogenesis for 48 hours. Lumen areas per field were determined by tracing EC lumens using Metamorph software from time-lapse images at the indicated time points. The effects of exogenous addition of TIMPs 1-4 and GM6001 on EC lumen formation over the time course is shown, with the adjacent bar graph highlighting EC lumen area at the final 48-hour time point. n = 3 fields per time point.
Figure 2
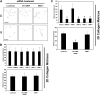
siRNAs directed to MT1-MMP block EC lumen formation and generation of vascular guidance tunnels in 3D collagen matrices. (A) The graph shows the average corresponding lumen area in micrometers per high-powered field for each siRNA treatment. Cultures were examined at either 24 or 48 hours of culture. The inability of MT1-MMP siRNA treated cells to form lumens is shown, with the bars representing the average lumenal area ± SD (P < .01; n = 3). (B) The graph shows the average corresponding vascular guidance tunnel area in micrometers per high-powered field for each siRNA treatment. Individual fields were photographed under fluorescence after immunostaining of gels (24-hour cultures) with anti-collagen type I antibodies. Vascular guidance tunnel areas were traced using Metamorph software from the photographs. The inability of MT1-MMP siRNA-treated cells to form tunnels is shown, with the bars representing average tunnel area per field ± SD (P < .01). (C) Western blots showing MT1-MMP expression demonstrate specific knockdown of the gene with siRNA directed to MT1-MMP compared with MT3-MMP and MMP-1 as well as the control siRNA directed to luciferase. Actin was used as a loading control. (D) Representative images of siRNA-transfected ECs seeded within FITC-labeled collagen type I 3D matrices are shown. Luciferase (Luc), MT1-MMP, MT3-MMP, and MMP-1 siRNA-transfected mRFP-ECs were allowed to form lumens and tube networks for 48 hours and data quantified (A). Bar equals 100 μm.
Figure 3
TIMPs 2, 3, and 4 and siRNA suppression of MT1-MMP block EC motility in 3D collagen matrices but not on 2D collagen substrates. (A-C) For 2D assays, nuclear GFP-labeled ECs were seeded on collagen coated plastic while for 3D assays, nuclear GFP-labeled ECs were placed into collagen gels. Time-lapse fluorescence microscopy was used to track cell motion using nuclei as a measure of EC migratory events. GM6001 was added to the culture media at 5 μM while the TIMPs were used at 5 μg/mL in the media. siRNA knockdown was performed in ECs for MT1-MMP, MMP-1, and Luciferase as control. n = 25 cells quantitated for each condition. P < .01 compared with control. (A) Representative overlays of tracking data are shown. The images show the movement of single cells after siRNA treatment on 2D collagen surfaces versus within 3D collagen matrices. Bar equals 100 μm.
Figure 4
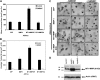
Increased expression of catalytically active but not inactive MT1-MMP increases EC lumen and vascular guidance tunnel formation. (A) ECs transfected with the indicated adenovirus were cultured for 24 hours, fixed, stained, and photographed. Adenoviral vectors used were control GFP, MMP-1, wild-type MT1-MMP (MT1-MMPWT), and mutant MT1-MMP (E240A) to inactivate the catalytic activity of the enzyme (MT1-MMPEA). Mean lumenal area in micrometers was measured, with bars representing area ± SD (P < .01). Blockade of this response is seen in all cell types by the addition of 5 μM GM6001, a broad-spectrum chemical MMP inhibitor. (B) Mean vascular guidance tunnel area was measured in micrometers, with bars representing area ± SD (P < .01). Blockade of this response is seen in all cell treatments after the addition of 5 μM GM6001, a broad-spectrum chemical MMP inhibitor. (C) Increased expression of MT1-MMP but not other proteins leads to increased EC lumenal area. Cultures were fixed and stained after 24 hours and photographed. Bar equals 50 μm. (D) Western blot analysis demonstrates increased expression of MT1-MMP in ECs after adenoviral infection with AdMT1-MMP but not other adenoviruses.
Figure 5
Vascular guidance tunnels are physical spaces in 3D collagen matrices that are generated by MT1-MMP-mediated proteolysis during EC tube morphogenesis, which support EC motility and vascular tube remodeling. (A) ECs were seeded within a 3D collagen matrix and allowed to form vascular tube networks for 48 hours. (B) Vascular networks were rapidly regressed for 10 minutes with the phosphatase inhibitor calyculin A. Arrows indicate borders of vascular guidance tunnels; arrowheads indicate collapsed EC aggregates. Bar equals 25 μm. (C) Silicone oil was injected into vascular guidance tunnels from a single injection site using a 2-μm diameter micropipette (arrows). Cultures were allowed to form (A), were collapsed (B) with calyculin A, and were microinjected. A montage of photographs are shown from this extensive vascular guidance tunnel network present within the 3D collagen matrices (arrowheads) as a result of EC lumen and tube morphogenesis. Bar equals 20 μm. (D) Photographs from time-lapse images showing EC tube remodeling (denoted by white arrows) whereby ECs and tube structures are observed moving through vascular guidance tunnels (outlined by white arrowheads). Black arrows indicate process extension and tube movement into a vacated vascular guidance tunnel space (white arrowheads). Bar equals 25 μm.
Figure 6
EC motility within vascular guidance tunnels is an MMP-independent process. Nuclear GFP-ECs were allowed to undergo morphogenesis in 3D collagen matrices for 48 hours to establish a network of tubes and vascular guidance tunnels. At 48 hours, GM6001 was either added or not to block further proteolysis and time-lapse imaging done. (A) Tracking of nuclei was done and representative tracings of cellular movement are shown. (B-C) Quantification of EC total distance traveled and average velocity are shown, demonstrating that EC migration within vascular guidance tunnels does not depend on MMP activity.
Figure 7
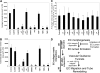
Inhibitors of EC lumen formation block vascular guidance tunnel formation, and differential role of integrins in controlling EC motility and adhesive interactions with vascular guidance tunnel matrices. ECs were seeded into collagen matrices, and various inhibitors of lumen formation were added from the beginning of the assay as indicated. The indicated drugs (Go6976, Go6983, PP2) were each added at 10 μM, the integrin blocking antibodies were added at 20 μg/mL, and the MMP inhibitors, GM6001, and the recombinant TIMPs were added at 5 μM and 5 μg/mL, respectively. (A) Average luminal area was measured in micrometers from 5 independent cultures, with bars representing area ± SD (P < .01) and quantitated from images obtained from stained cultures. (B) Cultures were immunostained for collagen type I and quantification of average vascular guidance tunnel formation measured in micrometers from 5 independent cultures, with bars representing tunnel area ± SD (P < .01). (C) Nuclear GFP-EC cultures were established for 48 hours, after which time integrin blocking antibodies were added at 20 μg/mL and real-time imaging was performed to assess EC motility within vascular guidance tunnels. Velocity of migration was quantitated from 20 independent cell motility tracings from triplicate cultures and is calculated as μm per minute. (D) Schematic diagram showing that EC morphogenic processes lead to both lumen formation and vascular guidance tunnel formation. The lumen formation mechanism depends on MT1-MMP-dependent proteolysis and the α2β1 integrin in a 3D matrix environment. ECs initially are completely surrounded by collagen matrix. Vascular guidance tunnels which form as a consequence of EC lumen formation are then used as 2D migratory matrix surfaces allowing EC motility and tube remodeling events that are MT1-MMP-independent. The ECs flatten out within these tunnel spaces and are interacting with collagen ECM on their abluminal surfaces while their luminal surfaces are exposed to fluid, thus, mimicking a 2D matrix environment. EC migratory events involve αv and α2β1 integrins that recognize the proteolytically altered vascular guidance tunnel matrix surface (containing both native and denatured collagen type I).
Similar articles
- Endothelial lumen signaling complexes control 3D matrix-specific tubulogenesis through interdependent Cdc42- and MT1-MMP-mediated events.
Sacharidou A, Koh W, Stratman AN, Mayo AM, Fisher KE, Davis GE. Sacharidou A, et al. Blood. 2010 Jun 24;115(25):5259-69. doi: 10.1182/blood-2009-11-252692. Epub 2010 Mar 9. Blood. 2010. PMID: 20215637 Free PMC article. - Mechanisms controlling human endothelial lumen formation and tube assembly in three-dimensional extracellular matrices.
Davis GE, Koh W, Stratman AN. Davis GE, et al. Birth Defects Res C Embryo Today. 2007 Dec;81(4):270-85. doi: 10.1002/bdrc.20107. Birth Defects Res C Embryo Today. 2007. PMID: 18228260 Review. - Molecular mechanisms controlling vascular lumen formation in three-dimensional extracellular matrices.
Sacharidou A, Stratman AN, Davis GE. Sacharidou A, et al. Cells Tissues Organs. 2012;195(1-2):122-43. doi: 10.1159/000331410. Epub 2011 Oct 13. Cells Tissues Organs. 2012. PMID: 21997121 Free PMC article. Review. - Molecular basis for endothelial lumen formation and tubulogenesis during vasculogenesis and angiogenic sprouting.
Davis GE, Stratman AN, Sacharidou A, Koh W. Davis GE, et al. Int Rev Cell Mol Biol. 2011;288:101-65. doi: 10.1016/B978-0-12-386041-5.00003-0. Int Rev Cell Mol Biol. 2011. PMID: 21482411 Free PMC article. Review. - MT1-MMP- and Cdc42-dependent signaling co-regulate cell invasion and tunnel formation in 3D collagen matrices.
Fisher KE, Sacharidou A, Stratman AN, Mayo AM, Fisher SB, Mahan RD, Davis MJ, Davis GE. Fisher KE, et al. J Cell Sci. 2009 Dec 15;122(Pt 24):4558-69. doi: 10.1242/jcs.050724. Epub 2009 Nov 24. J Cell Sci. 2009. PMID: 19934222 Free PMC article.
Cited by
- Matrix metalloproteinases: inflammatory regulators of cell behaviors in vascular formation and remodeling.
Chen Q, Jin M, Yang F, Zhu J, Xiao Q, Zhang L. Chen Q, et al. Mediators Inflamm. 2013;2013:928315. doi: 10.1155/2013/928315. Epub 2013 Jun 12. Mediators Inflamm. 2013. PMID: 23840100 Free PMC article. Review. - Controlled activation of morphogenesis to generate a functional human microvasculature in a synthetic matrix.
Hanjaya-Putra D, Bose V, Shen YI, Yee J, Khetan S, Fox-Talbot K, Steenbergen C, Burdick JA, Gerecht S. Hanjaya-Putra D, et al. Blood. 2011 Jul 21;118(3):804-15. doi: 10.1182/blood-2010-12-327338. Epub 2011 Apr 28. Blood. 2011. PMID: 21527523 Free PMC article. - Mature and progenitor endothelial cells perform angiogenesis also under protease inhibition: the amoeboid angiogenesis.
Chillà A, Margheri F, Biagioni A, Del Rosso M, Fibbi G, Laurenzana A. Chillà A, et al. J Exp Clin Cancer Res. 2018 Apr 3;37(1):74. doi: 10.1186/s13046-018-0742-2. J Exp Clin Cancer Res. 2018. PMID: 29615071 Free PMC article. - Biophysiochemical properties of endothelial cells cultured on bio-inspired collagen films.
Seo E, Seo KW, Gil JE, Ha YR, Yeom E, Lee S, Lee SJ. Seo E, et al. BMC Biotechnol. 2014 Jul 2;14:61. doi: 10.1186/1472-6750-14-61. BMC Biotechnol. 2014. PMID: 24984812 Free PMC article.
References
- Adams RH, Alitalo K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol. 2007;8:464–478. - PubMed
- Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005;438:932–936. - PubMed
- Davis GE, Bayless KJ, Mavila A. Molecular basis of endothelial cell morphogenesis in three-dimensional extracellular matrices. Anat Rec. 2002;268:252–275. - PubMed
- Davis GE, Saunders WB. Molecular balance of capillary tube formation versus regression in wound repair: role of matrix metalloproteinases and their inhibitors. J Invest Dermatol Symp Proc. 2006;11:44–56. - PubMed
- Drake CJ. Embryonic and adult vasculogenesis. Birth Defects Res C Embryo Today. 2003;69:73–82. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL079460/HL/NHLBI NIH HHS/United States
- R01 HL059373/HL/NHLBI NIH HHS/United States
- R01 HL087308/HL/NHLBI NIH HHS/United States
- R21 HL085659/HL/NHLBI NIH HHS/United States
- R01 HL070308/HL/NHLBI NIH HHS/United States
- HL59373/HL/NHLBI NIH HHS/United States
- HL79460/HL/NHLBI NIH HHS/United States
- HL87308/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous