Uterine-specific p53 deficiency confers premature uterine senescence and promotes preterm birth in mice - PubMed (original) (raw)
Uterine-specific p53 deficiency confers premature uterine senescence and promotes preterm birth in mice
Yasushi Hirota et al. J Clin Invest. 2010 Mar.
Abstract
Many signaling pathways that contribute to tumorigenesis are also functional in pregnancy, although they are dysregulated in the former and tightly regulated in the latter. Transformation-related protein 53 (Trp53), which encodes p53, is a tumor suppressor gene whose mutation is strongly associated with cancer. However, its role in normal physiological processes, including female reproduction, is poorly understood. Mice that have a constitutive deletion of Trp53 exhibit widespread development of carcinogenesis at early reproductive ages, compromised spermatogenesis, and fetal exencephaly, rendering them less amenable to studying the role of p53 in reproduction. To overcome this obstacle, we generated mice that harbor a conditional deletion of uterine Trp53 and examined pregnancy outcome in females with this genotype. These mice had normal ovulation, fertilization, and implantation; however, postimplantation uterine decidual cells showed terminal differentiation and senescence-associated growth restriction with increased levels of phosphorylated Akt and p21, factors that are both known to participate in these processes in other systems. Strikingly, uterine deletion of Trp53 increased the incidence of preterm birth, a condition that was corrected by oral administration of the selective COX2 inhibitor celecoxib. We further generated evidence to suggest that deletion of uterine Trp53 induces preterm birth through a COX2/PGF synthase/PGF(2alpha) pathway. Taken together, our observations underscore what we believe to be a new critical role of uterine p53 in parturition.
Figures
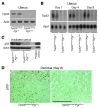
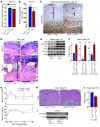
Figure 1. Efficient deletion of uterine Trp53.
(A and B) Trp53 mRNA was undetectable in 2-month-old Trp53loxP/loxPPgrCre/+ uteri analyzed by RT-PCR (A) and Northern hybridization (B). Actb and Rpl7 were used as internal controls. (C) p53 protein levels were very low to undetectable in irradiation-induced 2-month-old Trp53loxP/loxPPgrCre/+ uteri assessed by Western blotting. Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ dams were killed 6 hours after 9.5 Gy whole-body γ irradiation. Protein extracts from mouse lymphoma cell line WR19L were used as a positive control. (D) Immunostaining of p53 in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ decidua on day 8 of pregnancy. Immunoreactive p53 was undetectable in Trp53loxP/loxPPgrCre/+ decidua. Fast Green solution was used to counterstain the cytoplasm. Dark green-black staining shows nuclear p53 localization. Scale bar: 200 μm.
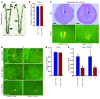
Figure 2. Mice with uterine deletion of Trp53 show normal implantation.
(A) Representative photographs of uteri with implantation sites (blue bands) on day 5 of pregnancy in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ females, mated with fertile Trp53loxP/loxPPgr+/+ males. Arrows and arrowheads indicate implantation sites and ovaries, respectively. (B) Number of implantation sites on day 5 of pregnancy (mean ± SEM; P > 0.05; Student’s t test). (C) Representative photomicrographs of H&E-stained histology, and in situ hybridization of Ptgs2, in implantation sites on day 5 of pregnancy in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ dams. Arrowheads indicate the location of implanting blastocysts. Scale bars: 200 μm. (D) In situ hybridization of Lif in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ uterine sections on days 1 and 4 of pregnancy. Scale bars: 200 μm. (E and F) Lif mRNA levels in day 1 uteri of Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ dams (E) and in day 4, 0900 h, uteri of Trp53loxP/loxPPgr+/+, Trp53loxP/loxPPgrCre/+, WT, and Lif+/– dams (F), as determined by Northern hybridization. Values are mean ± SEM of 2–4 independent samples. *P < 0.05, Student’s t tests.
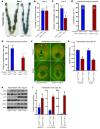
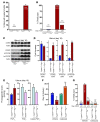
Figure 3. Uterine deletion of Trp53 restricts normal decidual growth.
(A) Representative photographs of day 8 pregnant implantation sites in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ mice. (B and C) Number and weight of implantation sites in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ mice on day 8 of pregnancy (mean ± SEM; *P < 0.05). (D and E) Trp53loxP/loxPPgr+/+ blastocysts were transferred into uteri of Trp53loxP/loxPPgr+/+ or Trp53loxP/loxPPgrCre/+ recipients on day 4 of pseudopregnancy. The number and weight of implantation sites were evaluated on day 8, 4 days after embryo transfer (mean ± SEM; *P < 0.05). Numbers in parentheses denote the ratio of implantation sites/total blastocysts transferred. (F) In situ hybridization of Hoxa10 and Bmp2 in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ uteri on day 8 of pregnancy. Em, embryo; M, mesometrial pole; AM, antimesometrial pole. Scale bar: 200 μm. (G) Northern hybridization of Hoxa10 and Bmp2 in day 8 uteri of Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ dams. In each group, 2–3 independent samples were examined (mean ± SEM; *P < 0.05). (H and I) Loss of p53 upregulated pAkt, p110α, and p21 levels in day 8 implantation sites. As assessed by Western blotting (H), quantitatively analyzed band intensities of pAkt were normalized against total Akt, and those of p110α and p21 were normalized against actin (I). In each group, 3 independent samples from different mice were examined (mean ± SEM; *P < 0.05).
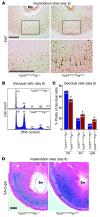
Figure 4. Uterine deletion of Trp53 confers decidual polyploidy, terminal differentiation, and senescence.
(A) Uterine loss of p53 increased Ki67-positive decidual cells with large nuclei. Higher-magnification views of the boxed regions are shown below. Arrowheads indicate Ki67-positive cells with large, irregularly shaped nuclei. Scale bars: 200 μm. (B) Representative histogram of flow cytometry analyses showing DNA content (propidium iodide). Ploidy is denoted by -N (i.e., 2N, diploid; 4N–10N, polyploid). (C) In Trp53loxP/loxPPgrCre/+ decidua, the number of diploid cells decreased, whereas the number of polyploid cells increased (mean ± SEM; *P < 0.05, Student’s t tests). (D) Uterine Trp53 deletion enhanced SA–β-gal activity in decidua. Scale bar: 200 μm.
Figure 5. Decidual senescence persists in uteri deleted of Trp53.
(A and B) Number and weight of implantation sites in Trp53loxP/loxPPgr+/+ and Trp53loxP/loxPPgrCre/+ mice on day 16 of pregnancy (mean ± SEM; *P < 0.05). (**C**) Uterine loss of p53 impeded decidual growth. Placental labyrinth and spongiotrophoblast layers were demarcated by cytokeratin-8 immunostaining. (**D**) Uterine _Trp53_ deletion enhanced SA–β-gal activity primarily in the decidual layer on days 12 and 16. (**C** and **D**) Decidual boundaries are demarcated by double-sided arrows. Myo, myometrium; Dec, decidua; Sp, spongiotrophoblast; Lb, labyrinth; BV, blood vessel. Scale bars: 500 μm. (**E** and **F**) Levels of p21, pAkt, and p110α were upregulated in day 16 uteri lacking p53. (**E**) Uterine samples from which placentas and fetuses had been removed were used for Western blotting. (**F**) Quantitative analysis of band intensities of pAkt were normalized against total Akt, and those of p21 and p110α were normalized against actin (mean ± SEM; *_P_ < 0.05). In each group, 3 independent samples from different mice were examined. (**G**) Serum levels of E2 and P4 (mean ± SEM; _P_ > 0.05). At least 4 independent samples were examined in each group. (H) Representative photomicrographs of H&E-stained histological sections of ovaries on day 16 of pregnancy. CL, corpus luteum; F, follicle. Scale bar: 200 μm. (I) Western blotting of ovarian 20α-HSD levels on day 16 of pregnancy. Day-20 WT mice were used as a positive control. (J) Quantitative analysis of 20α-HSD band intensities from I were normalized against actin (mean ± SEM; P > 0.05).
Figure 6. Uterine deletion of Trp53 promotes preterm birth through COX2/PGFS/PGF2α pathway.
(A) Preterm birth (deliveries earlier than day 19) occurred in 52% of Trp53loxP/loxPPgrCre/+ dams (*P < 0.05). Numbers in parentheses denote the ratio of dams with preterm birth/total dams. (B) All pups born before day 19 were dead around the time of delivery or died immediately after birth (*P < 0.05 versus Trp53loxP/loxPPgr+/+). Numbers in parentheses denote the ratio of dead pups/total pups. (C and D) Levels of COX2 and PGFS were upregulated in day 16 uteri lacking p53. (C) Uterine samples from which placentas and fetuses had been removed were used for Western blotting, and (D) quantitative analysis of band intensities were all normalized against actin (mean ± SEM of 3 independent samples from different mice; *P < 0.05). (E) Conditional loss of uterine p53 upregulated PGF2α levels in day 16 uteri. A total of 15 independent samples from 3 different mice were evaluated (mean ± SEM; *P < 0.05). (F and G) Preterm birth and neonatal deaths in Trp53loxP/loxPPgrCre/+ mice were reversed by oral gavage of celecoxib (10 mg/kg/dose, administered twice) on day 16. In G, numbers in parentheses denote the ratio of dead pups/total pups. *P < 0.05 versus vehicle-treated Trp53loxP/loxPPgr+/+; **P < 0.05 versus vehicle-treated Trp53loxP/loxPPgrCre/+.
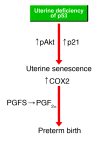
Figure 7. Potential pathways that promote uterine senescence and preterm birth in Trp53loxP/loxPPgrCre/+ mice.
Similar articles
- Combinatory approaches prevent preterm birth profoundly exacerbated by gene-environment interactions.
Cha J, Bartos A, Egashira M, Haraguchi H, Saito-Fujita T, Leishman E, Bradshaw H, Dey SK, Hirota Y. Cha J, et al. J Clin Invest. 2013 Sep;123(9):4063-75. doi: 10.1172/JCI70098. Epub 2013 Aug 27. J Clin Invest. 2013. PMID: 23979163 Free PMC article. - Uterine deletion of Trp53 compromises antioxidant responses in the mouse decidua.
Burnum KE, Hirota Y, Baker ES, Yoshie M, Ibrahim YM, Monroe ME, Anderson GA, Smith RD, Daikoku T, Dey SK. Burnum KE, et al. Endocrinology. 2012 Sep;153(9):4568-79. doi: 10.1210/en.2012-1335. Epub 2012 Jul 3. Endocrinology. 2012. PMID: 22759378 Free PMC article. - Evaluation of the tocolytic effect of a selective cyclooxygenase-2 inhibitor in a mouse model of lipopolysaccharide-induced preterm delivery.
Sakai M, Tanebe K, Sasaki Y, Momma K, Yoneda S, Saito S. Sakai M, et al. Mol Hum Reprod. 2001 Jun;7(6):595-602. doi: 10.1093/molehr/7.6.595. Mol Hum Reprod. 2001. PMID: 11385116 - The role of decidual cells in uterine hemostasis, menstruation, inflammation, adverse pregnancy outcomes and abnormal uterine bleeding.
Schatz F, Guzeloglu-Kayisli O, Arlier S, Kayisli UA, Lockwood CJ. Schatz F, et al. Hum Reprod Update. 2016 Jun;22(4):497-515. doi: 10.1093/humupd/dmw004. Epub 2016 Feb 23. Hum Reprod Update. 2016. PMID: 26912000 Free PMC article. Review. - Is there a role for placental senescence in the genesis of obstetric complications and fetal growth restriction?
Sultana Z, Maiti K, Dedman L, Smith R. Sultana Z, et al. Am J Obstet Gynecol. 2018 Feb;218(2S):S762-S773. doi: 10.1016/j.ajog.2017.11.567. Epub 2017 Dec 22. Am J Obstet Gynecol. 2018. PMID: 29275823 Review.
Cited by
- Molecular Regulation of Parturition: The Role of the Decidual Clock.
Norwitz ER, Bonney EA, Snegovskikh VV, Williams MA, Phillippe M, Park JS, Abrahams VM. Norwitz ER, et al. Cold Spring Harb Perspect Med. 2015 Apr 27;5(11):a023143. doi: 10.1101/cshperspect.a023143. Cold Spring Harb Perspect Med. 2015. PMID: 25918180 Free PMC article. Review. - Comparison of the fertility of tumor suppressor gene-deficient C57BL/6 mouse strains reveals stable reproductive aging and novel pleiotropic gene.
Kohzaki M, Ootsuyama A, Umata T, Okazaki R. Kohzaki M, et al. Sci Rep. 2021 Jun 11;11(1):12357. doi: 10.1038/s41598-021-91342-9. Sci Rep. 2021. PMID: 34117297 Free PMC article. - Tumor promoter-induced cellular senescence: cell cycle arrest followed by geroconversion.
Leontieva OV, Blagosklonny MV. Leontieva OV, et al. Oncotarget. 2014 Dec 30;5(24):12715-27. doi: 10.18632/oncotarget.3011. Oncotarget. 2014. PMID: 25587030 Free PMC article. - The Regulators of Human Endometrial Stromal Cell Decidualization.
Murata H, Tanaka S, Okada H. Murata H, et al. Biomolecules. 2022 Sep 10;12(9):1275. doi: 10.3390/biom12091275. Biomolecules. 2022. PMID: 36139114 Free PMC article. Review. - Candidate gene linkage approach to identify DNA variants that predispose to preterm birth.
Bream EN, Leppellere CR, Cooper ME, Dagle JM, Merrill DC, Christensen K, Simhan HN, Fong CT, Hallman M, Muglia LJ, Marazita ML, Murray JC. Bream EN, et al. Pediatr Res. 2013 Feb;73(2):135-41. doi: 10.1038/pr.2012.166. Epub 2012 Nov 20. Pediatr Res. 2013. PMID: 23168575 Free PMC article.
References
- Behrman RE, Butler AS, eds.Preterm Birth. Washington, DC: The National Academies Press; 2007. - PubMed
- Wang H, Dey SK. Roadmap to embryo implantation: clues from mouse models. Nat Rev Genet. 2006;7(3):185–199. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R37 HD012304/HD/NICHD NIH HHS/United States
- DA01822/DA/NIDA NIH HHS/United States
- HD12304/HD/NICHD NIH HHS/United States
- R01 DA006668/DA/NIDA NIH HHS/United States
- R37 DA006668/DA/NIDA NIH HHS/United States
- P30 AR047363/AR/NIAMS NIH HHS/United States
- AR47363/AR/NIAMS NIH HHS/United States
- DA006668/DA/NIDA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous