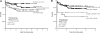Natural history of transient myeloproliferative disorder clinically diagnosed in Down syndrome neonates: a report from the Children's Oncology Group Study A2971 - PubMed (original) (raw)
Clinical Trial
. 2011 Dec 22;118(26):6752-9; quiz 6996.
doi: 10.1182/blood-2011-04-350017. Epub 2011 Aug 17.
Todd A Alonzo, Robert B Gerbing, Joanne M Hilden, April D Sorrell, Mukta Sharma, Thomas W Loew, Robert J Arceci, Dorothy Barnard, John Doyle, Gita Massey, John Perentesis, Yaddanapudi Ravindranath, Jeffrey Taub, Franklin O Smith
Affiliations
- PMID: 21849481
- PMCID: PMC3245202
- DOI: 10.1182/blood-2011-04-350017
Clinical Trial
Natural history of transient myeloproliferative disorder clinically diagnosed in Down syndrome neonates: a report from the Children's Oncology Group Study A2971
Alan S Gamis et al. Blood. 2011.
Abstract
Transient myeloproliferative disorder (TMD), restricted to newborns with trisomy 21, is a megakaryocytic leukemia that although lethal in some is distinguished by its spontaneous resolution. Later development of acute myeloid leukemia (AML) occurs in some. Prospective enrollment (n = 135) elucidated the natural history in Down syndrome (DS) patients diagnosed with TMD via the use of uniform monitoring and intervention guidelines. Prevalent at diagnosis were leukocytosis, peripheral blast exceeding marrow blast percentage, and hepatomegaly. Among those with life-threatening symptoms, most (n = 29/38; 76%) received intervention therapy until symptoms abated and then were monitored similarly. Organomegaly with cardiopulmonary compromise most frequently led to intervention (43%). Death occurred in 21% but only 10% were attributable to TMD (intervention vs observation patients: 13/14 vs 1/15 because of TMD). Among those solely observed, peripheral blasts and all other TMD symptoms cleared at a median of 36 and 49 days from diagnosis, respectively. On the basis of the diagnostic clinical findings of hepatomegaly with or without life-threatening symptoms, 3 groups were identified with differing survival: low risk with neither finding (38%), intermediate risk with hepatomegaly alone (40%), and high risk with both (21%; overall survival: 92% ± 8%, 77% ± 12%, and 51% ± 19%, respectively; P ≤ .001). Among all, AML subsequently occurred in 16% at a median of 441 days (range, 118-1085 days). The trial is registered at http://www.clinicaltrials.gov as NCT00003593.
Figures
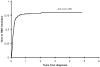
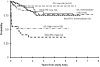
Figure 1
Time to TMD resolution from diagnosis for all patients enrolled.
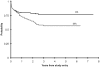
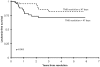
Figure 2
OS and EFS from study entry for all TMD patients.
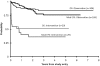
Figure 3
OS and mEFS (Mod EFS) from study entry comparing observation to intervention patients. Deaths that are not TMD related are competing events in modified EFS (1 − cumulative incidence).
Figure 4
OS and modified EFS (mEFS) based on diagnostic hepatomegaly and WBC values. (A) Diagnostic hepatomegaly. (B) WBC values. These illustrate the univariate impact upon OS and modified EFS of these 2 risk factors.
Figure 5
OS and modified EFS (Mod EFS) for the 3 risk groups of TMD patients. This illustrates that the intermediate risk group, that is, those with hepatomegaly alone, had minimal TMD-related problems but did have significant mortality from other causes.
Figure 6
Impact of TMD resolution upon later risk of developing AML. This illustrates that those children whose resolution of their TMD exceeded the median resolution time were at greater risk of later developing AML.
Comment in
- Enigmatic variation.
Roberts I, Vyas P. Roberts I, et al. Blood. 2011 Dec 22;118(26):6723-4. doi: 10.1182/blood-2011-09-376145. Blood. 2011. PMID: 22194390 No abstract available.
Similar articles
- [GATA1-mutation associated leukemia in children with trisomy 21 mosaic].
Reinhardt D, Reinhardt K, Neuhoff C, Sander A, Klusmann JH, Pekrun A, Sauerbrey A, von Stackelberg A, Rössig C, Creutzig U, Kolenova A. Reinhardt D, et al. Klin Padiatr. 2012 Apr;224(3):153-5. doi: 10.1055/s-0032-1308988. Epub 2012 Apr 18. Klin Padiatr. 2012. PMID: 22513796 German. - Development of acute megakaryoblastic leukemia from a minor clone in a Down syndrome patient with clinically overt transient myeloproliferative disorder.
Xu G, Kato K, Toki T, Takahashi Y, Terui K, Ito E. Xu G, et al. J Pediatr Hematol Oncol. 2006 Oct;28(10):696-8. doi: 10.1097/01.mph.0000212997.02554.f6. J Pediatr Hematol Oncol. 2006. PMID: 17023834 - Megakaryoblastic disorders in children.
Lorsbach RB. Lorsbach RB. Am J Clin Pathol. 2004 Dec;122 Suppl:S33-46. doi: 10.1309/Y57UGTE36PGQ2NV6. Am J Clin Pathol. 2004. PMID: 15690641 Review. - Acute nonlymphocytic leukemia after transient myeloproliferative disorder in a patient with Down syndrome.
Barnett PL, Clark AC, Garson OM. Barnett PL, et al. Med Pediatr Oncol. 1990;18(5):347-53. doi: 10.1002/mpo.2950180502. Med Pediatr Oncol. 1990. PMID: 2142749 Review.
Cited by
- Down syndrome-associated leukaemias: current evidence and challenges.
Mason NR, Cahill H, Diamond Y, McCleary K, Kotecha RS, Marshall GM, Mateos MK. Mason NR, et al. Ther Adv Hematol. 2024 Jul 23;15:20406207241257901. doi: 10.1177/20406207241257901. eCollection 2024. Ther Adv Hematol. 2024. PMID: 39050114 Free PMC article. Review. - GATA1 in Normal and Pathologic Megakaryopoiesis and Platelet Development.
Takasaki K, Chou ST. Takasaki K, et al. Adv Exp Med Biol. 2024;1459:261-287. doi: 10.1007/978-3-031-62731-6_12. Adv Exp Med Biol. 2024. PMID: 39017848 Review. - Cytokine profiling in 128 patients with transient abnormal myelopoiesis: a report from the JPLSG TAM-10 trial.
Yamato G, Tsumura Y, Muramatsu H, Shimada A, Imaizumi T, Tsukagoshi H, Kaburagi T, Shiba N, Yamada Y, Deguchi T, Kawai T, Terui K, Ito E, Watanabe K, Hayashi Y. Yamato G, et al. Blood Adv. 2024 Jun 25;8(12):3120-3129. doi: 10.1182/bloodadvances.2023011628. Blood Adv. 2024. PMID: 38691583 Free PMC article. Clinical Trial. - Transient Abnormal Myelopoiesis: An Abnormal Course and the Efficacy of Delayed Treatment.
Mishra P, Fajrudheen M, Sahoo T, Som TK, Biswal S, Chhabra G. Mishra P, et al. Cureus. 2024 Feb 14;16(2):e54219. doi: 10.7759/cureus.54219. eCollection 2024 Feb. Cureus. 2024. PMID: 38496060 Free PMC article. - Too many white cells-TAM, JMML, or something else?
Satty A, Stieglitz E, Kucine N. Satty A, et al. Hematology Am Soc Hematol Educ Program. 2023 Dec 8;2023(1):37-42. doi: 10.1182/hematology.2023000464. Hematology Am Soc Hematol Educ Program. 2023. PMID: 38066851 Free PMC article.
References
- Gamis AS, Hilden JM. Transient myeloproliferative disorder, a disorder with too few data and many unanswered questions: does it contain an important piece of the puzzle to understanding hematopoiesis and acute myelogenous leukemia? J Pediatr Hematol Oncol. 2002;24(1):2–5. - PubMed
- Kurahashi H, Hara J, Yumura-Yagi K, et al. Monoclonal nature of transient abnormal myelopoiesis in Down's syndrome. Blood. 1991;77(6):1161–1163. - PubMed
- Zipursky A, Brown EJ, Christensen H, Doyle J. Transient myeloproliferative disorder (transient leukemia) and hematologic manifestations of Down syndrome. Clin Lab Med. 1999;19(1):157–67. vii. - PubMed
- Pine S, Guo Q, Yin C, Jayabose S, Druschel C, Sandoval C. Incidence and clinical implications of GATA1 mutations in newborns with Down syndrome. Blood. 2007;110:2128–2131. - PubMed
- Zipursky A, Brown E, Christensen H, Sutherland R, Doyle J. Leukemia and/or myeloproliferative syndrome in neonates with Down syndrome. Semin Perinatol. 1997;21(1):97–101. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials