Bone erosion in rheumatoid arthritis: mechanisms, diagnosis and treatment - PubMed (original) (raw)
Review
Bone erosion in rheumatoid arthritis: mechanisms, diagnosis and treatment
Georg Schett et al. Nat Rev Rheumatol. 2012 Nov.
Abstract
Bone erosion is a central feature of rheumatoid arthritis and is associated with disease severity and poor functional outcome. Erosion of periarticular cortical bone, the typical feature observed on plain radiographs in patients with rheumatoid arthritis, results from excessive local bone resorption and inadequate bone formation. The main triggers of articular bone erosion are synovitis, including the production of proinflammatory cytokines and receptor activator of nuclear factor κB ligand (RANKL), as well as antibodies directed against citrullinated proteins. Indeed, both cytokines and autoantibodies stimulate the differentiation of bone-resorbing osteoclasts, thereby stimulating local bone resorption. Although current antirheumatic therapy inhibits both bone erosion and inflammation, repair of existing bone lesions, albeit physiologically feasible, occurs rarely. Lack of repair is due, at least in part, to active suppression of bone formation by proinflammatory cytokines. This Review summarizes the substantial progress that has been made in understanding the pathophysiology of bone erosions and discusses the improvements in the diagnosis, monitoring and treatment of such lesions.
Figures
Figure 1
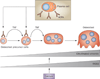
Autoantibodies against citrullinated proteins and osteoclastogenesis. Plasma cells produce ACPA with specificity for citrullinated vimentin, which bind to osteoclast precursor cells and stimulate the release of TNF, which in turn enhances the differentiation of these cells into mature osteoclasts. During the osteoclast differentiation process, production of the PADI2 enzyme is induced by calcium. The activity of PADI2 leads to citrullination of vimentin, which is abundantly expressed on the surface of osteoclast-lineage cells. Abbreviations: ACPA, anti-citrullinated protein antibodies; PADI2, peptidyl-arginine deiminase type 2.
Figure 2
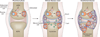
Evolution of bone erosion in the course of RA. During the preclinical phase of RA, ACPA are produced early on by plasma cells. ACPA can stimulate osteoclast differentiation and lead to initial bone loss. These early changes may initiate in the bone marrow adjacent to the joint. Synovitis at the onset of clinical disease leads to production of cytokines, which stimulate osteoclastogenesis by inducing expression of RANKL, and synergize with RANKL to enhance bone erosion. Established RA is characterized by the presence of large bone erosions filled with inflamed, synovially derived pannus tissue. Abbreviations: ACPA, anti-citrullinated protein antibodies; RA, rheumatoid arthritis; RANKL, receptor activator of nuclear factor κB ligand.
Figure 3
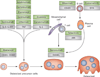
Site of action of antirheumatic drugs on osteoclast differentiation and bone erosion. Inhibitors (green boxes) of TNF, IL-1 and IL-6R block the expression of RANKL by mesenchymal cells and T cells; they also directly interfere with osteoclastogenesis. Abatacept inhibits osteoclast differentiation by directly engaging CD80 and CD86 on the surface of osteoclast precursor cells. T-cell activation is targeted by small-molecule tyrosine kinase inhibitors such as tofacitinib, an inhibitor of JAK. B cells differentiate into plasma cells, which are a source of RANKL. B cells are depleted by an antibody against CD20 (rituximab) and are inhibited by small-molecule tyrosine kinase inhibitors such as fostamatinib, an inhibitor of SYK. Abbreviations: IL-6R, IL-6 receptor; JAK, Janus kinase; RANKL, receptor activator of nuclear factor κB ligand; SYK, spleen tyrosine kinase.
Figure 4
Disruption of bone homeostasis by synovitis. Inflammation within synovial tissue induces osteoclastogenesis through increased expression of RANKL, and by the production of proinflammatory cytokines that drive osteoclastogenesis and synergize with RANKL. In addition, expression of Dkk-1 by synovial fibroblasts leads to inhibition of osteoblast differentiation and consequently of bone formation. Dkk-1 itself induces expression of another anti-anabolic molecule, sclerostin, by osteocytes. Abbreviations: Dkk-1; Dickkopf-related protein 1; RANKL, receptor activator of nuclear factor κB ligand.
Similar articles
- Rheumatoid Arthritis in the View of Osteoimmunology.
Auréal M, Machuca-Gayet I, Coury F. Auréal M, et al. Biomolecules. 2020 Dec 31;11(1):48. doi: 10.3390/biom11010048. Biomolecules. 2020. PMID: 33396412 Free PMC article. Review. - Pathogenesis of bone erosions in rheumatoid arthritis.
Goldring SR, Gravallese EM. Goldring SR, et al. Curr Opin Rheumatol. 2000 May;12(3):195-9. doi: 10.1097/00002281-200005000-00006. Curr Opin Rheumatol. 2000. PMID: 10803748 Review. - Fc-gamma receptors and S100A8/A9 cause bone erosion during rheumatoid arthritis. Do they act as partners in crime?
Di Ceglie I, Kruisbergen NNL, van den Bosch MHJ, van Lent PLEM. Di Ceglie I, et al. Rheumatology (Oxford). 2019 Aug 1;58(8):1331-1343. doi: 10.1093/rheumatology/kez218. Rheumatology (Oxford). 2019. PMID: 31180451 Review. - Bone and joint destruction in rheumatoid arthritis: what is really happening?
Goldring SR. Goldring SR. J Rheumatol Suppl. 2002 Sep;65:44-8. J Rheumatol Suppl. 2002. PMID: 12236623 Review. - [Inflammation and osteoclasts].
Yokota K. Yokota K. Nihon Rinsho Meneki Gakkai Kaishi. 2017;40(5):367-376. doi: 10.2177/jsci.40.367. Nihon Rinsho Meneki Gakkai Kaishi. 2017. PMID: 29238019 Review. Japanese.
Cited by
- ROCK inhibition with Y-27632 reduces joint inflammation and damage in serum-induced arthritis model and decreases in vitro osteoclastogenesis in patients with early arthritis.
Rodríguez-Trillo A, Pena C, García S, Pérez-Pampín E, Rodríguez-López M, Mera-Varela A, González A, Conde C. Rodríguez-Trillo A, et al. Front Immunol. 2022 Aug 11;13:858069. doi: 10.3389/fimmu.2022.858069. eCollection 2022. Front Immunol. 2022. PMID: 36032152 Free PMC article. - Regulatory role of KCa3.1 in immune cell function and its emerging association with rheumatoid arthritis.
Lin Y, Zhao YJ, Zhang HL, Hao WJ, Zhu RD, Wang Y, Hu W, Zhou RP. Lin Y, et al. Front Immunol. 2022 Oct 5;13:997621. doi: 10.3389/fimmu.2022.997621. eCollection 2022. Front Immunol. 2022. PMID: 36275686 Free PMC article. Review. - Plasma Sclerostin Levels in Rheumatoid Arthritis Women on TNF-α Inhibitor Therapy.
Szeremeta A, Jura-Półtorak A, Zoń-Giebel A, Olczyk K, Komosińska-Vassev K. Szeremeta A, et al. Pharmaceuticals (Basel). 2024 May 22;17(6):666. doi: 10.3390/ph17060666. Pharmaceuticals (Basel). 2024. PMID: 38931334 Free PMC article. - Tocilizumab decreases T cells but not macrophages in the synovium of patients with rheumatoid arthritis while it increases the levels of serum interleukin-6 and RANKL.
Chatzidionysiou K, Circiumaru A, Rethi B, Joshua V, Engstrom M, Hensvold A, Af Klint E, Catrina A. Chatzidionysiou K, et al. RMD Open. 2021 Jun;7(2):e001662. doi: 10.1136/rmdopen-2021-001662. RMD Open. 2021. PMID: 34112702 Free PMC article. - Calcitonin treatment for osteoarthritis and rheumatoid arthritis - a systematic review and meta-analysis of preclinical data.
Meyer Günderoth M, Bannach-Brown A, Winkler T, Keller J, Zahn RK, Maleitzke T. Meyer Günderoth M, et al. EFORT Open Rev. 2024 Jul 1;9(7):600-614. doi: 10.1530/EOR-23-0133. EFORT Open Rev. 2024. PMID: 38949173 Free PMC article.
References
- Karsenty G, Kronenberg HM, Settembre C. Genetic control of bone formation. Annu. Rev. Cell. Dev. Biol. 2009;25:629–648. - PubMed
- Teitelbaum SL, Ross FP. Genetic regulation of osteoclast development and function. Nat. Rev. Genet. 2003;4:638–649. - PubMed
- Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–342. - PubMed
- Baker WM. The formation of abnormal synovial cysts in the connection with the joints. St Bartolomews Hospital Reports. 1855;21:177–190.
- Weichselbaum A. Die feineren Veränderungen des Gelenkknorpels bei fungöser Synovitis und Karies der Gelenkenden [German] Archiv. Pathol. Anat. Physiol. Klin. Med. 1878;73:461–475.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical