Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of α-synuclein in Parkinson's disease models - PubMed (original) (raw)
. 2013 Aug 15;22(16):3315-28.
doi: 10.1093/hmg/ddt192. Epub 2013 May 10.
Affiliations
- PMID: 23666528
- PMCID: PMC3723316
- DOI: 10.1093/hmg/ddt192
Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of α-synuclein in Parkinson's disease models
Michaeline L Hebron et al. Hum Mol Genet. 2013.
Erratum in
- Correction to: Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of α-synuclein in Parkinson's disease models.
[No authors listed] [No authors listed] Hum Mol Genet. 2023 Jan 1;32(1):172-176. doi: 10.1093/hmg/ddac274. Hum Mol Genet. 2023. PMID: 36370466 Free PMC article. No abstract available.
Abstract
Parkinson's disease is a movement disorder characterized by death of dopaminergic substantia nigra (SN) neurons and brain accumulation of α-synuclein. The tyrosine kinase Abl is activated in neurodegeneration. Here, we show that lentiviral expression of α-synuclein in the mouse SN leads to Abl activation (phosphorylation) and lentiviral Abl expression increases α-synuclein levels, in agreement with Abl activation in PD brains. Administration of the tyrosine kinase inhibitor nilotinib decreases Abl activity and ameliorates autophagic clearance of α-synuclein in transgenic and lentiviral gene transfer models. Subcellular fractionation shows accumulation of α-synuclein and hyper-phosphorylated Tau (p-Tau) in autophagic vacuoles in α-synuclein expressing brains, but nilotinib enhances protein deposition into the lysosomes. Nilotinib is used for adult leukemia treatment and it enters the brain within US Food and Drug Administration approved doses, leading to autophagic degradation of α-synuclein, protection of SN neurons and amelioration of motor performance. These data suggest that nilotinib may be a therapeutic strategy to degrade α-synuclein in PD and other α-synucleinopathies.
Figures
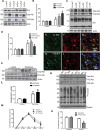
Figure 1.
Abl activation is associated with accumulation of α-synuclein. WB on 10% SDS-NuPAGE gel shows (A) lentiviral α-synuclein expression (first blot), total Abl (second blot) and tyrosine 412 (T412) phosphorylated Abl (third blot) relative to actin (n = 9) and (B) graphs represent densitometry analysis. (C) Total Abl (first blot) and tyrosine 412 (T412) phosphorylated Abl (second blot), and mouse α-synuclein expression (third blot) relative to actin (n = 9) in lentiviral Abl- and LacZ-injected mice, and (D) graphs represent densitometry analysis. IHC in 20 μm thick brain sections showing (E) V5, (F) Abl and (G) merged V5 and Abl staining in the SN of mice injected with lentiviral Abl. IHC in 20 μm thick brain sections showing (H) V5, (I) Abl and (J) merged V5 and Abl staining in the SN of mice injected with lentiviral LacZ. WB on 4–12% SDS-NuPAGE gel shows (K) total Abl (first blot) and T412 Abl (second blot) relative to actin in human post-mortem striatal extracts, n = 9 PD and 7 controls, P < 0.02, two-tailed _t_-test, and (L) densitometry of human WBs. (M) Graph represents quantification of mass spectroscopy analysis of brain nilotinib (n = 5/time point). (N) WB on 4–12% SDS-NuPAGE gel shows total Abl (first blot), T412 Abl (second blot) T245 Abl (third blot) and phospho-tyrosine (fourth blot) relative to parkin in wild-type mice injected with DMSO or nilotinib once daily for 3 weeks and (O) graphs represent densitometry analysis. *Significantly different, ANOVA, Neumann–Keuls multiple comparison, P < 0.05. n = number of animals, and bars are means.
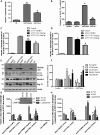
Figure 2.
Abl inhibition via nilotinib promotes autophagic degradation of α-synuclein. Graphs represent ELISA measurement of (A) human α-synuclein (n = 14) and (B) caspase-3 activity (n = 64) in 6–8-month-old transgenic A53T mice and wild-type age-matched controls injected daily i.p. with 10 mg/kg nilotinib for 3 weeks. Graphs represent ELISA measurement of (A) brain levels of human α-synuclein (n = 10) and (B) blood levels of human α-synuclein (n = 10) in 5-month-old transgenic A53T mice and wild-type age-matched controls treated i.p. with 5 mg/kg or 1 mg/kg nilotinib every other day for 6 weeks. WB analysis on 4–12% SDS-NuPAGE gel shows (E) monomeric and high molecular weight human α-synuclein (first blot), LC3 (second blot), Beclin-1 (third blot) and Atg12 (fourth blot) relative to actin (n = 9) in A53T mice treated daily i.p. with 10 mg/kg nilotinib for 3 weeks, and (F) densitometry analysis. Graphs represent ELISA measurement of (G) human α-synuclein (insert shows WB of AVs) and (H) p-Tau levels in A53T mice (n = 5) treated daily i.p. with 10 mg/kg nilotinib for 3 weeks. *Significantly different, ANOVA, Neumann–Keuls multiple comparison, P < 0.05. n = number of animals, bars are means.
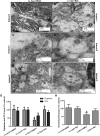
Figure 3.
Nilotinib attenuates α-synuclein levels in A53T mice. (A) An MRI scan of a mouse brain showing brain orientation in subsequent images. Immunohistochemical staining of 20 μm thick brain sections shows (B) no loss of TH in VTA and SN in 6–8-month-old A53T mice, and abundant expression of human α-synuclein in (C) the striatum of DMSO-treated A53T mice, and (D) striatum of nilotinib-treated A53T mice, (E) the cortex of DMSO-treated A53T mice, (F) cortex of nilotinib-treated A53T mice, (G) the hippocampus of DMSO-treated A53T mice, and (H) hippocampus of nilotinib-treated A53T mice. The images represent different animals with daily i.p. injection of nilotinib for 3 weeks.
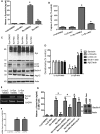
Figure 4.
Nilotinib attenuates α-synuclein levels in the SN of gene transfer mice. Graphs represent ELISA measurement in mesencephalic extracts showing (A) human α-synuclein (n = 9) and (B) caspase-3 activity (n = 14) in 6-month-old C57BL/6 mice injected with lentiviral α-synuclein or LacZ into the SN for 3 weeks and treated daily i.p. with 10 mg/kg nilotinib for three additional weeks. WB analysis on 4–12% SDS-NuPAGE gel shows (C) monomeric and high molecular weight mouse α-synuclein (first blot), LC3 (second blot), Beclin-1 (third blot) and Atg12 (fourth blot) relative to actin (n = 9) in 6-month-old C57BL/6 mice injected with lentiviral α-synuclein or LacZ into the SN for 3 weeks and treated daily i.p. with 10 mg/kg nilotinib for three additional weeks, and (D) densitometry analysis. (E) Gels represent the levels of human α-synuclein mRNA in 6-month-old C57BL/6 mice injected with lentiviral α-synuclein or LacZ into the SN for 3 weeks and treated daily i.p. with 10 mg/kg nilotinib for three additional weeks and (F) graphs represent quantification of RT-PCR results relative to GADPH. (G) ELISA of human α-synuclein in M17 neuroblastoma cells transfected with 3 μg LacZ, α-synuclein cDNA or Beclin-1 shRNA for 24 h and then treated with 10 μ
m
nilotinib for additional 24 h with and without bafilomycin A1 (n = 12). *Significantly different, ANOVA, Neumann–Keuls multiple comparison, P < 0.05. n = number of animals or number of cell culture experiments. Bars are means.
Figure 5.
Nilotinib clears α-synuclein and protects TH+ neurons. Immunohistochemical staining of 20 μm thick brain sections shows human α-synuclein in the SN of (A) lentiviral LacZ injected mice treated with 10 mg/kg nilotinib, (B) lentiviral α-synuclein treated with DMSO and (C) lentiviral α-synuclein treated with 10 mg/kg nilotinib daily for 3 weeks. Immunohistochemical staining of 20 μm thick brain sections show TH in (D) lentiviral LacZ injected with nilotinib, and (G) is higher magnification from a different animal, (E) lentiviral α-synuclein and treated with DMSO, and (H) is higher magnification from a different animal. (F) Lentiviral α-synuclein treated with nilotinib, and (I) is higher magnification from a different animal. High magnification of (J) TH+ neurons and (M) Nissl-stained sections in lentiviral LacZ treated with DMSO. High magnification of (K) TH+ neurons and (N) Nissl-stained sections in lentiviral α-synuclein treated with DMSO. High magnification of (L) TH+ neurons and (O) Nissl-stained sections in lentiviral α-synuclein treated with 10 mg/kg nilotinib for 3 weeks.
Figure 6.
Nilotinib clears AVs, increases DA level and improves motor performance in SN of lentiviral α-synuclein mice. Transmission electron microscopy of SN neurons shows (A, C, E) accumulation of cytosolic debris and AVs in lentiviral α-synuclein expressing mice treated with DMSO. (B, D, F) Appearance of larger AVs with different levels of maturation in lentiviral α-synuclein treated with 10 mg/kg nilotinib for 3 weeks. Graphs represent (G) DA and HVA ELISA levels in mesencephalon brain extracts of lentiviral α-synuclein treated with DMSO compared with LacZ mice (n = 8) and (H) shows time spent (%) on rotarod in lentiviral α-synuclein or LacZ injected mice with and without nilotinib (n = 14). *Significantly different, ANOVA, Neumann–Keuls multiple comparison, P < 0.05. n = number of animals, bars are means.
Comment in
- Tyrosine kinase inhibition facilitates autophagic SNCA/α-synuclein clearance.
Hebron ML, Lonskaya I, Moussa CE. Hebron ML, et al. Autophagy. 2013 Aug;9(8):1249-50. doi: 10.4161/auto.25368. Epub 2013 Jun 19. Autophagy. 2013. PMID: 23787811 Free PMC article.
Similar articles
- Tyrosine kinase inhibition facilitates autophagic SNCA/α-synuclein clearance.
Hebron ML, Lonskaya I, Moussa CE. Hebron ML, et al. Autophagy. 2013 Aug;9(8):1249-50. doi: 10.4161/auto.25368. Epub 2013 Jun 19. Autophagy. 2013. PMID: 23787811 Free PMC article. - α-Synucleinopathy associated c-Abl activation causes p53-dependent autophagy impairment.
Karim MR, Liao EE, Kim J, Meints J, Martinez HM, Pletnikova O, Troncoso JC, Lee MK. Karim MR, et al. Mol Neurodegener. 2020 Apr 16;15(1):27. doi: 10.1186/s13024-020-00364-w. Mol Neurodegener. 2020. PMID: 32299471 Free PMC article. - Caffeic acid reduces A53T α-synuclein by activating JNK/Bcl-2-mediated autophagy in vitro and improves behaviour and protects dopaminergic neurons in a mouse model of Parkinson's disease.
Zhang Y, Wu Q, Zhang L, Wang Q, Yang Z, Liu J, Feng L. Zhang Y, et al. Pharmacol Res. 2019 Dec;150:104538. doi: 10.1016/j.phrs.2019.104538. Epub 2019 Nov 9. Pharmacol Res. 2019. PMID: 31707034 - Parkinson's Disease Modification Through Abl Kinase Inhibition: An Opportunity.
Werner MH, Olanow CW. Werner MH, et al. Mov Disord. 2022 Jan;37(1):6-15. doi: 10.1002/mds.28858. Epub 2021 Nov 23. Mov Disord. 2022. PMID: 34816484 Free PMC article. Review. - The usual suspects, dopamine and alpha-synuclein, conspire to cause neurodegeneration.
Mor DE, Daniels MJ, Ischiropoulos H. Mor DE, et al. Mov Disord. 2019 Feb;34(2):167-179. doi: 10.1002/mds.27607. Epub 2019 Jan 11. Mov Disord. 2019. PMID: 30633814 Free PMC article. Review.
Cited by
- Nilotinib - Differentiating the Hope from the Hype.
Wyse RK, Brundin P, Sherer TB. Wyse RK, et al. J Parkinsons Dis. 2016 Jul 12;6(3):519-22. doi: 10.3233/JPD-160904. J Parkinsons Dis. 2016. PMID: 27434298 Free PMC article. - Tyrosine kinases: multifaceted receptors at the intersection of several neurodegenerative disease-associated processes.
Stevenson M, Algarzae NK, Moussa C. Stevenson M, et al. Front Dement. 2024 Aug 16;3:1458038. doi: 10.3389/frdem.2024.1458038. eCollection 2024. Front Dement. 2024. PMID: 39221072 Free PMC article. Review. - Lysosomal dysfunction in α-synuclein pathology: molecular mechanisms and therapeutic strategies.
Dai L, Liu M, Ke W, Chen L, Fang X, Zhang Z. Dai L, et al. Cell Mol Life Sci. 2024 Sep 3;81(1):382. doi: 10.1007/s00018-024-05419-5. Cell Mol Life Sci. 2024. PMID: 39223418 Free PMC article. Review. - Drug Repurposing in Parkinson's Disease.
Athauda D, Foltynie T. Athauda D, et al. CNS Drugs. 2018 Aug;32(8):747-761. doi: 10.1007/s40263-018-0548-y. CNS Drugs. 2018. PMID: 30066310 Review. - Therapeutics in the Pipeline Targeting _α_-Synuclein for Parkinson's Disease.
Grosso Jasutkar H, Oh SE, Mouradian MM. Grosso Jasutkar H, et al. Pharmacol Rev. 2022 Jan;74(1):207-237. doi: 10.1124/pharmrev.120.000133. Pharmacol Rev. 2022. PMID: 35017177 Free PMC article. Review.
References
- Benner E.J., Banerjee R., Reynolds A.D., Sherman S., Pisarev V.M., Tsiperson V., Nemachek C., Ciborowski P., Przedborski S., Mosley R.L., et al. Nitrated alpha-synuclein immunity accelerates degeneration of nigral dopaminergic neurons. PLoS ONE. 2008;3:e1376. doi:10.1371/journal.pone.0001376. - DOI - PMC - PubMed
- Kuhn D.M., Francescutti-Verbeem D.M., Thomas D.M. Dopamine quinones activate microglia and induce a neurotoxic gene expression profile: relationship to methamphetamine-induced nerve ending damage. Ann. N. Y. Acad. Sci. 2006;1074:31–41. doi:10.1196/annals.1369.003. - DOI - PubMed
- Reynolds A.D., Kadiu I., Garg S.K., Glanzer J.G., Nordgren T., Ciborowski P., Banerjee R., Gendelman H.E. Nitrated alpha-synuclein and microglial neuroregulatory activities. J. Neuroimmune Pharmacol. 2008;3:59–74. doi:10.1007/s11481-008-9100-z. - DOI - PMC - PubMed
- Goedert M. Filamentous nerve cell inclusions in neurodegenerative diseases: tauopathies and alpha-synucleinopathies. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1999;354:1101–1118. doi:10.1098/rstb.1999.0466. - DOI - PMC - PubMed
- Goedert M. Alpha-synuclein and neurodegenerative diseases. Nat. Rev. Neurosci. 2001;2:492–501. doi:10.1038/35081564. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous