Localization-dependent and -independent roles of SLX4 in regulating telomeres - PubMed (original) (raw)
Localization-dependent and -independent roles of SLX4 in regulating telomeres
Jamie S J Wilson et al. Cell Rep. 2013.
Abstract
SLX4, a scaffold for structure-specific DNA repair nucleases, is important for several types of DNA repair. Many repair proteins bind to sites of DNA damage, resulting in subnuclear "foci," but SLX4 forms foci in human cells even without DNA damage. Using several approaches, we show that most, but not all, SLX4 foci localize to telomeres in a range of human cell lines irrespective of the mechanisms used to maintain telomere length. The SLX1 Holliday-junction-processing enzyme is recruited to telomeres by SLX4, and SLX4, in turn, is recruited by a motif that binds to the shelterin subunit TRF2 directly. We also show that TRF2-dependent recruitment of SLX4 prevents telomere damage. Furthermore, SLX4 prevents telomere lengthening and fragility in a manner that appears to be independent of telomere association. These findings reveal that SLX4 plays multiple roles in regulating telomere homeostasis.
Copyright © 2013 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
Graphical abstract
Figure 1
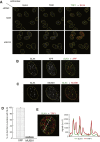
Endogenous SLX4 Complex Localizes at Telomeres in U2OS cells (A) Indirect immunofluorescence analysis of SLX4 and TRF2 in U2OS cells. Cells were fixed and preextracted, and SLX4 or TRF2 foci were visualized. (B) U2OS cells were fixed and SLX4 or TRF1 foci were visualized by indirect immunofluorescence. Cells were then fixed again and subjected to FISH analysis with a telomeric PNA probe. (C) Same as (A), except that a straight line was drawn using OMERO Insight (Allan et al., 2012) through a single Z-section of the nucleus, and the intensity of the SLX4, TRF2, or DAPI signals was quantitated along the length of the line. a.u.f., arbitrary units of fluorescence. (D) U2OS cells stably expressing GFP-SLX4 or an SLX4 mutant incapable of interacting with SLX1 (GFP-SLX4-C1805R) were fixed, and GFP-SLX4 or SLX1 foci were visualized by indirect immunofluorescence. (E) Indirect immunofluorescence analysis of SLX1 and TRF2 in U2OS cells stably overexpressing GFP-tagged SLX1. See also Figure S1.
Figure 2
Multiple Approaches Detect SLX4 at Telomeres in Several Human Cell Lines (A and B) U2OS cells were fixed with formaldehyde, and ChIP analysis was performed with the antibodies indicated. DNA from immunoprecipitates was subjected to 3-fold serial dilutions before spotting onto Hybond N+ and subjected to hybridization with a radioactively labeled telomeric probe or an Alu DNA probe. Input DNA prepared from cell extracts before immunoprecipitation was subjected to similar analysis. (C) ChIP analysis (left panels) and indirect immunofluorescence analysis of SLX4 or TRF2 foci (middle and right panels) in A549 cells. In the middle panel, a straight line was drawn through a single Z-section of the nucleus, and the intensity of the SLX4, TRF2, or DAPI signals was quantitated along the length of the line (right panel). (D) Same as (C) except that HeLa cells were used. a.u.f., arbitrary units of fluorescence. In all ChIP experiments, the “input DNA” lane shows 10%, 3%, and 1% of the total DNA in cell extract, respectively. See also Figure S2.
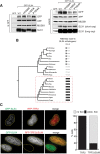
Figure 3
A TRF2-Binding Motif in SLX4 (A) Yeast two hybrid analysis was performed with a GAL4 activation domain (GAD) fusion of SLX4 and GAL4 DNA binding domain (GBD) fusion of TRF2, RAP1, or XPF to detect interaction between these proteins. Cells grown on medium lacking LEU and TRP (to select for bait and prey plasmids) were replica plated to medium lacking LEU, TRP, and HIS to test for activation of the HIS3 reporter gene. (B) Schematic diagram of the modular domain organization of SLX4. The putative TBM is highlighted in red, and the sequence of the putative TBM from SLX4 is compared with the classical TRF2-binding motif (TBM). (C) U2OS cells were cotransfected with RFP-tagged TRF2 and HA-tagged SLX4 wild-type (WT), or HA-SLX4 bearing alanine substitutions at H1020, L1022, or P1024. Vector expressing HA tag only was used as control. Cells were lysed and subjected to immunoprecipitation with anti-RFP antibodies, and precipitates were probed with the antibodies indicated. (D) Same as (C) except that SLX4 was GFP tagged, and extracts were subjected to immunoprecipitation with anti-GFP antibodies. (E) Indirect immunofluorescence analysis of U2OS cells stably expressing GFP-SLX4-L1022A. GFP-SLX4 or endogenous TRF2 foci were visualized. (F) The proportion of foci in U2OS cells formed by GFP-SLX4 or GFP-SLX4-L1022A that cocolocalize with TRF2, that are adjacent to but not overlapping with TRF2, or that do not colocalize with TRF2 was quantitated. (G) Telomere-ChIP analysis of GFP-SLX4 or GFP-SLX4-L1022A, or GFP only, stably expressed in U2OS cells. DNA from immunoprecipitates was subjected to 3-fold serial dilutions, before spotting onto Hybond N+ and hybridization with a radioactively labeled telomeric probe (upper panel) or an Alu DNA probe (lower panel). Input DNA prepared from cell extracts before immunoprecipitation was subjected to similar analysis. The “input DNA” lane shows 10%, 3%, and 1% of the total DNA in cell extract, respectively. (H) The ChIP signal in the dots corresponding to each serial dilution for each immunoprecipitate in (G) was quantitated and added together. To normalize the hybridization signals, the resulting totals for each precipitate were divided by the total input signal (left panels). See also Figures S2 and S3.
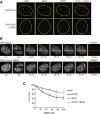
Figure 4
Localization-Dependent and -Independent Roles of SLX4 in Regulating Telomere Homeostasis (A) U2OS cells stably expressing tetracycline-inducible forms of GFP-SLX4 wild-type (WT) or GFP-SLX4-L1022A were incubated, or not, with tetracycline for 48 hr to induce maximal protein expression. Cells were fixed and subjected to immunofluorescence analysis of 53BP1 foci and TRF1 foci. The proportion of cells with greater than three 53BP1 foci that colocalized with TRF1 (“telomeric”), and that did not colocalize with TRF1 (“nontelomeric”) was counted before and after addition of tetracycline. The fold change after addition of tetracycline was calculated. At least 200 cells were counted for each condition, in two independent experiments. (B) Telomere fluorescence distribution of individual telomere dots in paraffin-embedded liver sections of mice of the indicated genotype as determined by q-FISH analysis. Mean fluorescence is indicated by the red horizontal line; a.u.f., arbitrary units of fluorescence. The total numbers of mice and nuclei analyzed per genotype are indicated. The Wilcoxon-Mann-Whitney rank sum test was used for statistical comparison; p values are indicated. (C) Telomere fluorescence distribution of individual telomere dots in MEFs from wild-type mice or _Slx4_−/− mice stably expressing mouse SLX4, as determined by q-FISH analysis. Empty vector was used as control. Mean fluorescence is indicated by the red horizontal line; a.u.f., arbitrary units of fluorescence. The total numbers of metaphase spreads (“meta”), and telomeres (“telo”) analyzed per genotype are indicated. (D) Frequency of multitelomeric signals (MTSs) per metaphase in primary MEFs of the indicated genotypes. Data are presented as mean and SEM. Representative images of MTSs are shown. The Wilcoxon-Mann-Whitney rank sum test was used for statistical comparison; p values are indicated. See also Figures S3 and S4.
Figure S1
Foci Formed by SLX4-Associated Nucleases, Related to Figure 1 (A) U2OS cells transfected with an SLX4-specific siRNA (SLX4-4) were fixed and endogenous SLX4 foci were visualized by indirect immunofluorescence using antibodies raised against amino acids 381-680 of human SLX4. TRF1 foci were also visualized. (B and C) Indirect immunofluorescence analysis of SLX4 and XPF (B), or SLX4 and MUS81 (C), in U2OS cells. Cells were fixed and the relevant foci were visualized. (D) Quantitation of the proportion of U2OS cells in which MUS81 or XPF foci colocalize with SLX4. (E) CCL-211 primary lung fibroblasts were fixed and SLX4 or TRF1 foci were visualized by indirect immunofluorescence. A straight line was drawn using OMERO Insight (Allan et al., 2012) through a single Z-section of the nucleus and the intensity of the SLX4 or TRF1 signals were quantitated along the length of the line. a.u.f, arbitrary units of fluorescence.
Figure S2
Effect of the SLX4 L1022A TBM Mutation on SLX4-Associated Nucleases, Related to Figure 3 (A) HEK293 cells were transiently transfected with vectors expressing GFP only, GFP-SLX4 wild-type (WT) or GFP-SLX4 bearing alanine mutations at H1020, L1022 or P1024. Extracts were subjected to Western blot analysis (right panels), or immunoprecipitation with GFP-Trap beads (left panels) followed by Western blotting. (B) Dendrogram of SLX4 orthologs from the species indicated. The diagram shows the TRF2-binding motifs (TBM) in SLX4 orthologs in primate species (boxed with a red dotted line), and the most similar motifs in a selection of other mammals. (C) U2OS cells stably expressing GFP-SLX4 were transiently transfected with plasmids encoding RFP-tagged TRF2 or TRF2ΔBΔM. After 48 hr, cells were fixed and RFP-TRF2 and GFP-SLX4 foci were visualized (left panels). Quantitation of these data are shown in the right panel.
Figure S3
Effect of the SLX4 L1022A TBM Mutation on DNA Repair and on SLX4 Localization at Sites of DNA Damage, Related to Figure 4 (A) Indirect immunofluorescence analysis of fixed U2OS cells stably expressing GFP-SLX4 wild-type or GFP-SLX4-L1022A. GFP, SLX1 or TRF2 foci were visualized. (B) Live-cell fluorescent images of U2OS cells stably expressing GFP-SLX4 or GFP-SLX4-L1022A. Images were obtained at the indicated times after laser-micro-irradiation. The extreme right-hand panels show immunofluorescence images of γ-H2AX in cells fixed 60 min after irradiation. (C) Cells from Fanconi anemia patient EUFA-1354 were stably transfected with constructs expressing SLX4, SLX4-L1022A or empty vector. Cells were exposed to the indicated doses of mitomycin-C (MMC) for 18 hr, and after 72 hr cell viability was assessed by the MTS assay. Viability of untreated cells was defined as 100%. Data are represented as mean ± s.e.m, n = 3.
Figure S4
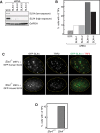
Depletion or Deletion of SLX4 from Mammalian Cells Causes Telomeric DNA Damage, Related to Figure 4 (A) U2OS cells were transfected with four different SLX4-specific siRNA duplexes and after 48 hr, SLX4 expression was tested by Western blotting. A scrambled version of siRNA SLX4-1 was used as control. (B) U2OS Cells treated with the indicated siRNAs were fixed and subjected to immunofluorescence analysis of 53BP1 foci and TRF1 foci. The proportion of cells with greater than three 53BP1 foci that co-localized with TRF1 (TIFs) was counted in at least 200 cells, in two independent experiments. (C) Mouse SLX4 or human SLX4, tagged at the N-terminus with GFP, were expressed in MEFs from _Slx4_−/− mice. Cells were fixed and GFP-SLX4 and TRF2 foci were visualized by indirect immunofluorescence. (D) MEFs of the genotypes indicated were fixed and processed as described in (B). The proportion of cells with greater than 3 TIFs in at least 200 cells in two independent experiments.
Similar articles
- SLX4 assembles a telomere maintenance toolkit by bridging multiple endonucleases with telomeres.
Wan B, Yin J, Horvath K, Sarkar J, Chen Y, Wu J, Wan K, Lu J, Gu P, Yu EY, Lue NF, Chang S, Liu Y, Lei M. Wan B, et al. Cell Rep. 2013 Sep 12;4(5):861-9. doi: 10.1016/j.celrep.2013.08.017. Epub 2013 Sep 5. Cell Rep. 2013. PMID: 24012755 Free PMC article. - SLX4 contributes to telomere preservation and regulated processing of telomeric joint molecule intermediates.
Sarkar J, Wan B, Yin J, Vallabhaneni H, Horvath K, Kulikowicz T, Bohr VA, Zhang Y, Lei M, Liu Y. Sarkar J, et al. Nucleic Acids Res. 2015 Jul 13;43(12):5912-23. doi: 10.1093/nar/gkv522. Epub 2015 May 18. Nucleic Acids Res. 2015. PMID: 25990736 Free PMC article. - PARP1 modulates telomere sister chromatid exchange and telomere length homeostasis by regulating telomere localization of SLX4 in U2OS cells.
Sadhukhan R, Ghosh U. Sadhukhan R, et al. Life Sci. 2021 Jul 15;277:119556. doi: 10.1016/j.lfs.2021.119556. Epub 2021 May 1. Life Sci. 2021. PMID: 33945829 - Coordinated roles of SLX4 and MutSβ in DNA repair and the maintenance of genome stability.
Young SJ, West SC. Young SJ, et al. Crit Rev Biochem Mol Biol. 2021 Apr;56(2):157-177. doi: 10.1080/10409238.2021.1881433. Epub 2021 Feb 17. Crit Rev Biochem Mol Biol. 2021. PMID: 33596761 Free PMC article. Review. - SLX4: multitasking to maintain genome stability.
Guervilly JH, Gaillard PH. Guervilly JH, et al. Crit Rev Biochem Mol Biol. 2018 Oct;53(5):475-514. doi: 10.1080/10409238.2018.1488803. Epub 2018 Oct 4. Crit Rev Biochem Mol Biol. 2018. PMID: 30284473 Review.
Cited by
- Dimerization of SLX4 contributes to functioning of the SLX4-nuclease complex.
Yin J, Wan B, Sarkar J, Horvath K, Wu J, Chen Y, Cheng G, Wan K, Chin P, Lei M, Liu Y. Yin J, et al. Nucleic Acids Res. 2016 Jun 2;44(10):4871-80. doi: 10.1093/nar/gkw354. Epub 2016 Apr 29. Nucleic Acids Res. 2016. PMID: 27131364 Free PMC article. - RAD52 and SLX4 act nonepistatically to ensure telomere stability during alternative telomere lengthening.
Verma P, Dilley RL, Zhang T, Gyparaki MT, Li Y, Greenberg RA. Verma P, et al. Genes Dev. 2019 Feb 1;33(3-4):221-235. doi: 10.1101/gad.319723.118. Epub 2019 Jan 28. Genes Dev. 2019. PMID: 30692206 Free PMC article. - Alternative Lengthening of Telomeres: Building Bridges To Connect Chromosome Ends.
Hoang SM, O'Sullivan RJ. Hoang SM, et al. Trends Cancer. 2020 Mar;6(3):247-260. doi: 10.1016/j.trecan.2019.12.009. Epub 2020 Jan 23. Trends Cancer. 2020. PMID: 32101727 Free PMC article. Review. - Human cancer cells utilize mitotic DNA synthesis to resist replication stress at telomeres regardless of their telomere maintenance mechanism.
Özer Ö, Bhowmick R, Liu Y, Hickson ID. Özer Ö, et al. Oncotarget. 2018 Mar 23;9(22):15836-15846. doi: 10.18632/oncotarget.24745. eCollection 2018 Mar 23. Oncotarget. 2018. PMID: 29662610 Free PMC article.
References
- Blasco M.A. Telomere length, stem cells and aging. Nat. Chem. Biol. 2007;3:640–649. - PubMed
- Cesare A.J., Reddel R.R. Alternative lengthening of telomeres: models, mechanisms and implications. Nat. Rev. Genet. 2010;11:319–330. - PubMed
Supplemental References
Publication types
MeSH terms
Substances
Grants and funding
- WT_/Wellcome Trust/United Kingdom
- 232854/ERC_/European Research Council/International
- MC_U127070192/MRC_/Medical Research Council/United Kingdom
- MC_UU_12016/1/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous