Immunogenic Chemotherapy Sensitizes Tumors to Checkpoint Blockade Therapy - PubMed (original) (raw)
. 2016 Feb 16;44(2):343-54.
doi: 10.1016/j.immuni.2015.11.024. Epub 2016 Feb 9.
Camilla Engblom 2, Steffen Rickelt 3, Virna Cortez-Retamozo 1, Christopher Garris 2, Ferdinando Pucci 1, Takahiro Yamazaki 4, Vichnou Poirier-Colame 4, Andita Newton 1, Younes Redouane 1, Yi-Jang Lin 1, Gregory Wojtkiewicz 1, Yoshiko Iwamoto 1, Mari Mino-Kenudson 5, Tiffany G Huynh 5, Richard O Hynes 3, Gordon J Freeman 6, Guido Kroemer 4, Laurence Zitvogel 4, Ralph Weissleder 1, Mikael J Pittet 7
Affiliations
- PMID: 26872698
- PMCID: PMC4758865
- DOI: 10.1016/j.immuni.2015.11.024
Immunogenic Chemotherapy Sensitizes Tumors to Checkpoint Blockade Therapy
Christina Pfirschke et al. Immunity. 2016.
Abstract
Checkpoint blockade immunotherapies can be extraordinarily effective, but might benefit only the minority of patients whose tumors are pre-infiltrated by T cells. Here, using lung adenocarcinoma mouse models, including genetic models, we show that autochthonous tumors that lacked T cell infiltration and resisted current treatment options could be successfully sensitized to host antitumor T cell immunity when appropriately selected immunogenic drugs (e.g., oxaliplatin combined with cyclophosphamide for treatment against tumors expressing oncogenic Kras and lacking Trp53) were used. The antitumor response was triggered by direct drug actions on tumor cells, relied on innate immune sensing through toll-like receptor 4 signaling, and ultimately depended on CD8(+) T cell antitumor immunity. Furthermore, instigating tumor infiltration by T cells sensitized tumors to checkpoint inhibition and controlled cancer durably. These findings indicate that the proportion of cancers responding to checkpoint therapy can be feasibly and substantially expanded by combining checkpoint blockade with immunogenic drugs.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures
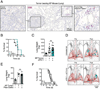
Figure 1. _Kras-Trp53_-mutated lung adenocarcinomas are inadequately infiltrated by T cells and resist current treatment options
A) Immunohistochemistry of CD3+ cells in KP lung tumor tissue on day 66 after tumor initiation. (B) Survival of KP mice treated or not with anti-PD-1 (αPD-1) mAbs (n=5-6 mice per group). Tumors were induced on day 0 by intratracheal intubation and inhalation (i.t.) of an adenovirus expressing Cre recombinase (AdCre). Mice were treated every third or fourth day with anti-PD-1 Abs intraperitoneally (i.p.) starting from day 60 to 86. (C) Lung weight as proxy for tumor burden (Cortez-Retamozo et al., 2012) measured on day 44 in mice bearing orthotopic KP1.9 tumors and treated or not with anti-PD-1 mAbs every third or forth day from day 25 to 42 after tumor cell injection (n=9-12 mice per group). (D) Micro-computed tomography of KP-OVA mice both pre- (day 122) and post-treatment (day 146) with no antibody (ø) or with anti-PD-1 and anti-CTLA-4 (αPD-1 + αCTLA-4) mAbs. Tumors were induced with a lentiviral vector containing OVA peptide sequences (LucOS) i.t. and mAb treatment was performed every second or third day from day 133 to 145. (E) Lung weight (n=4-5 mice per group) and (F) survival (n=11 mice per group) of KP mice treated or not with paclitaxel and carboplatin (Ptax-Carbo). Mice were treated once a week for three weeks starting on day 63 post i.t. tumor initiation and lungs analyzed three days after the last drug injection. For survival studies, Ptax-Carbo was injected i.p. once a week. ns, not significant. See also Figure S1.
Figure 2. Selected drugs induce KP tumor cell immune phenotypes
A) In vitro HMGB1 release by three KP tumor cell lines, generated from lung tissue of tumor-bearing KP mice, in response to various chemotherapeutic drug combinations as determined by ELISA (n=2-4 replicates). (B) HMGB1 release by tumor cell lines treated with Mitoxantrone (4 µM), Oxaliplatin (300 µM) and/or Mafosfamide at different concentrations (16.5, 33 or 50 µg/ml) (n=4 replicates). (C) Calreticulin exposure by tumor cell lines treated with defined drug combinations measured by flow cytometry (n=6 replicates). (D) HMGB1 immunohistochemistry (left) and scoring (right) in lung tumor nodules (n=39-48) of KP mice untreated (ø) or treated with Ptax-Carbo or Oxa-Cyc (See also Figure S2C for comparable images). Scale bars: 100 µm. ****P < 0.0001; ns, not significant; CRT, Calreticulin; Carbo, Carboplatin; Cyc, Cyclophosphamide; Dtax, Docetaxel; Maf, Mafosfamide; Mtx, Mitoxantrone; Oxa, Oxaliplatin; Ptax, Paclitaxel.
Figure 3. Drugs selected for their immunogenicity delay KP cancer progression
A) Lung tumor burden identification (left) and quantification (right) by hematoxylin and eosin (H&E) staining. Mice were treated with Ptax-Carbo or Oxa-Cyc after establishment of lung adenocarcinomas for a duration of 3 weeks. (B) Lung tumor detection by noninvasive magnetic resonance imaging both pre- and post-treatment as in (A) (left) and quantification of tumor progression, defined as delta tumor volume in mm3, in individual mice over time (right, n=5-6 mice per group). Red and green arrowheads show progressing and regressing tumor areas, respectively. *P < 0.05; ***P < 0.001; ns, not significant; Tx, treatment; Carbo, Carboplatin; Cyc, Cyclophosphamide; Oxa, Oxaliplatin; Ptax, Paclitaxel. See also Figure S3.
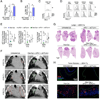
Figure 4. Drug-induced tumor control involves adaptive immunity
A and B) Impact of vaccinations with Ptax-Carbo- or Oxa-Maf-killed tumor cells on growth of KP1.9 tumors injected on day 0 ipsi- or contralaterally to the vaccination sites. Mice were injected with in vitro killed tumor cells on day −8, −4, −2, 5 and 12 (A) or only received prophylactic vaccination on day −8, −4 and −2 (B). Tumor burden was analyzed on day 19 (A) or day 14 (B), respectively (n=6 mice per group). (C) CD8+ T cell:Treg cell ratio in lungs of KP mice assessed by flow cytometry at 3 week post-treatment with Ptax-Carbo or Oxa-Cyc (n=9-13 mice per group). (D) CD3 immunohistochemistry of representative lung tumor sections from KP mice treated as in (C) (see Figure S4A for comparable images). Scale bars: 100 µm. (E) CD4 and CD8 immunohistochemistry of lung tumor tissue from Oxa-Cyc-treated KP mice (see Figure S4C for comparable images). Scale bars: 100 µm. (F) CD8+ T cell:Treg cell ratios assessed by flow cytometry in lung, bone marrow and spleen of KP1.9 lung tumor-bearing mice left untreated or that received Ptax-Carbo, Oxa, Cyc or Oxa-Cyc (n=7-8 mice per group). (G) Lung weight of Rag2−/− KP mice treated or not with Oxa-Cyc (n=12-14 mice per group). (H) Lung weight of Oxa-Cyc-treated KP mice that received CD8 depleting mAbs (αCD8, n=13-15 mice per group). *P < 0.05; **P < 0.01; ****P < 0.0001; ns, not significant. See also Figure S4.
Figure 5. Drug-induced tumor control involves innate immunity and TLR4 signaling
A) CD11b− and CD11b+ cells in lung tissue biopsies of KP mice that received Oxa-Cyc or were left untreated (n=7-8 mice per group). Lung tissue biopsies of tumor and tumor-free adjacent tissues were investigated in parallel. (B) TLR4 mean fluorescence intensity (MFI) of CD11b+ cell subsets in tumor and tumor-free lung tissues of Oxa-Cyc treated or untreated KP mice (n=7-8 mice per group). (C) CD103 phenotype of CD11b+CD11c− and CD11b+CD11c+ cells in tumor stroma of Oxa-Cyc-treated mice (n=7 mice per group). (D and E) Lung CD8+ T cell:Treg cell ratio (D) and lung weight (E) of KP1.9 tumor-bearing Tlr4+/+ and Tlr4−/− mice treated or not with Oxa-Cyc (n=7-14 mice per group). Lineage (Lin) defined as (B220/CD49b/CD90.2/Ter119)+. *P < 0.05; ***P < 0.001; ****P < 0.0001; ns, not significant; N/A, not applicable.
Figure 6. Drug-induced tumor infiltration by CD8+ T cells sensitizes lung adenocarcinomas to immune checkpoint therapy
A to D) Lung CD8+ T cell:Treg cell ratio (A), percent of OVA-specific CD8+ T cells in lungs (B), PD-1 expression by these cells (C) and PD-L1 surface expression by different tumor stroma cell populations (D; white histograms are fluorescence minus one (FMO) controls) in KP-OVA mice treated or not with Oxa-Cyc (n=2-5 mice per group). (E) Experimental scheme of micro-computed tomography imaging time points (d122, d146, d193) and ex vivo analysis (d234) of KP-OVA mice treated with Oxa-Cyc and anti-PD-1 + anti-CTLA-4 mAbs either alone or in combination (n=5 mice per group). Tumors were induced with a lentiviral vector containing OVA peptide sequences (LucOS). Change in tumor volume (defined by micro-computed tomography at d146 = T1 and d193 = T2) and tumor area in lung tissues (defined by H&E staining at d234 = T3) in these mice. (F) Coronal micro-computed tomography at d122, d146 and d193 of an untreated mouse (left) or mouse that received the combination therapy (right). Dotted lines identify tumor nodules that progressed (red) or not (green). (G) Lung tumor burden identification by H&E staining at d234 in the same mice. (H) CD8+ cell (red) infiltration in KP-OVA tumors (tumor contour defined with green dashed-lines, see Figure S5B for comparable images) identified by multiphoton microscopy ex vivo at d234 in the same mice. Collagen is shown in blue. *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant. See also Figure S5.
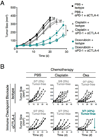
Figure 7. Immunogenic chemotherapeutics improve immune checkpoint blockade treatment against MCA205 fibrosarcoma and CT26 colon carcinoma
A) Tumor size measurement of MCA205 fibrosarcoma-bearing C57BL/6 mice (n=7-8 per group) treated with PBS or chemotherapy (Cisplatin or Doxorubicin) together with anti-PD-1 + anti-CTLA-4 (αPD-1 + αCTLA-4) mAbs or isotype control mAbs. Tumor cells were injected on d–8; the chemotherapeutics were given on d0 and the mAbs on d8, 12 and 16.. (B) Tumor size measurement of CT26 colon carcinoma-bearing BALB/c mice (n=7-8 per group) treated with PBS or chemotherapy (Cisplatin or Oxaliplatin (Oxa)) together with anti-CTLA-4 or isotype control mAbs. Tumor cells were injected on d–11; the chemotherapeutics were given on d0 and the mAbs on d8, 12 and 16. Each line represents an individual mouse. *P < 0.05; ns, not significant.
Similar articles
- Euphorbia Pekinensis Rupr. sensitizes colorectal cancer to PD-1 blockade by remodeling the tumor microenvironment and enhancing peripheral immunity.
Chen YY, Zeng XT, Gong ZC, Zhang MM, Wang KQ, Tang YP, Huang ZH. Chen YY, et al. Phytomedicine. 2024 Dec;135:156107. doi: 10.1016/j.phymed.2024.156107. Epub 2024 Sep 29. Phytomedicine. 2024. PMID: 39368338 - Improving PD-1 blockade plus chemotherapy for complete remission of lung cancer by nanoPDLIM2.
Sun F, Yan P, Xiao Y, Zhang H, Shapiro SD, Xiao G, Qu Z. Sun F, et al. Elife. 2024 Dec 24;12:RP89638. doi: 10.7554/eLife.89638. Elife. 2024. PMID: 39718207 Free PMC article. - Defining the optimum strategy for identifying adults and children with coeliac disease: systematic review and economic modelling.
Elwenspoek MM, Thom H, Sheppard AL, Keeney E, O'Donnell R, Jackson J, Roadevin C, Dawson S, Lane D, Stubbs J, Everitt H, Watson JC, Hay AD, Gillett P, Robins G, Jones HE, Mallett S, Whiting PF. Elwenspoek MM, et al. Health Technol Assess. 2022 Oct;26(44):1-310. doi: 10.3310/ZUCE8371. Health Technol Assess. 2022. PMID: 36321689 Free PMC article. - Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.
Salisbury L, Baraitser L. Salisbury L, et al. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review. - The effectiveness of abstinence-based and harm reduction-based interventions in reducing problematic substance use in adults who are experiencing homelessness in high income countries: A systematic review and meta-analysis: A systematic review.
O'Leary C, Ralphs R, Stevenson J, Smith A, Harrison J, Kiss Z, Armitage H. O'Leary C, et al. Campbell Syst Rev. 2024 Apr 21;20(2):e1396. doi: 10.1002/cl2.1396. eCollection 2024 Jun. Campbell Syst Rev. 2024. PMID: 38645303 Free PMC article. Review.
Cited by
- Tumor-intrinsic determinants of immunogenic cell death modalities.
Workenhe ST, Pol J, Kroemer G. Workenhe ST, et al. Oncoimmunology. 2021 Mar 2;10(1):1893466. doi: 10.1080/2162402X.2021.1893466. Oncoimmunology. 2021. PMID: 33717656 Free PMC article. Review. - Modulation of Determinant Factors to Improve Therapeutic Combinations with Immune Checkpoint Inhibitors.
Dosset M, Joseph EL, Rivera Vargas T, Apetoh L. Dosset M, et al. Cells. 2020 Jul 19;9(7):1727. doi: 10.3390/cells9071727. Cells. 2020. PMID: 32707692 Free PMC article. Review. - Chemokine switch regulated by TGF-β1 in cancer-associated fibroblast subsets determines the efficacy of chemo-immunotherapy.
Vienot A, Pallandre JR, Renaude E, Viot J, Bouard A, Spehner L, Kroemer M, Abdeljaoued S, van der Woning B, de Haard H, Loyon R, Hervouet E, Peixoto P, Borg C. Vienot A, et al. Oncoimmunology. 2022 Nov 12;11(1):2144669. doi: 10.1080/2162402X.2022.2144669. eCollection 2022. Oncoimmunology. 2022. PMID: 36387055 Free PMC article. - The Role of Tumor-Associated Myeloid Cells in Modulating Cancer Therapy.
Neophytou CM, Pierides C, Christodoulou MI, Costeas P, Kyriakou TC, Papageorgis P. Neophytou CM, et al. Front Oncol. 2020 Jun 9;10:899. doi: 10.3389/fonc.2020.00899. eCollection 2020. Front Oncol. 2020. PMID: 32656079 Free PMC article. Review. - Effectiveness and Safety of the Traditional Chinese Medicine Treatment (HuoxueHuayu Therapy) for Malignant Tumors: A Systematic Review and Meta-Analysis.
Chen Z, Wang A, Wei Y, Zhu Y, An J, Li Z. Chen Z, et al. Evid Based Complement Alternat Med. 2022 Aug 25;2022:7944063. doi: 10.1155/2022/7944063. eCollection 2022. Evid Based Complement Alternat Med. 2022. PMID: 38094221 Free PMC article. Review.
References
- Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E, Saulnier P, Yang H, Amigorena S, Ryffel B, Barrat FJ, Saftig P, Levi F, Lidereau R, Nogues C, Mira JP, Chompret A, Joulin V, Clavel-Chapelon F, Bourhis J, André F, Delaloge S, Tursz T, Kroemer G, Zitvogel L. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med. 2007;13:1050–1059. - PubMed
- Broz ML, Binnewies M, Boldajipour B, Nelson AE, Pollack JL, Erle DJ, Barczak A, Rosenblum MD, Daud A, Barber DL, Amigorena S, Van’t Veer LJ, Sperling AI, Wolf DM, Krummel MF. Dissecting the tumor myeloid compartment reveals rare activating antigen-presenting cells critical for T cell immunity. Cancer Cell. 2014;26:638–652. - PMC - PubMed
- Casanova JL, Abel L, Quintana-Murci L. Human TLRs and IL-1Rs in host defense: natural insights from evolutionary, epidemiological, and clinical genetics. Annu Rev Immunol. 2011;29:447–491. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 AI084880/AI/NIAID NIH HHS/United States
- P30 DK043351/DK/NIDDK NIH HHS/United States
- F31 CA196035/CA/NCI NIH HHS/United States
- P50 CA086355/CA/NCI NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- P30 CA014051/CA/NCI NIH HHS/United States
- 5U54-CA163109/CA/NCI NIH HHS/United States
- R01-AI084880/AI/NIAID NIH HHS/United States
- P50-CA86355/CA/NCI NIH HHS/United States
- R21 CA190344/CA/NCI NIH HHS/United States
- R01 CA204019/CA/NCI NIH HHS/United States
- U54 CA126515/CA/NCI NIH HHS/United States
- U54 CA163125/CA/NCI NIH HHS/United States
- 5U54CA163125/CA/NCI NIH HHS/United States
- U54-CA126515/CA/NCI NIH HHS/United States
- U54 CA163109/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous