Nucleoside - Definition and Examples - Biology Online Dictionary (original) (raw)
Nucleoside
n., plural: nucleosides
[xxxx]
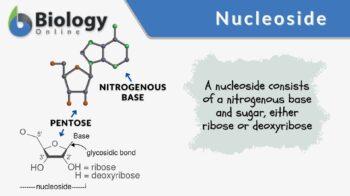
Definition: A nitrogenous base (purine or pyrimidine) bound to a pentose sugar ribose or deoxyribose
Table of Contents
Toggle
- Nucleoside Definition
- Characteristics of Nucleoside
- Types of Nucleoside
- Examples of Nucleosides
- Nucleoside Metabolism
- Biological Functions of Nucleosides
- Medical Importance
- Quiz
- Reference
A nucleoside is a nitrogenous base (purine or pyrimidine) bound to a pentose sugar ribose or deoxyribose. A nitrogenous base (also called nucleobase) is a nitrogen-containing compound that may form a nucleoside when they are attached to a five-carbon sugar ribose or deoxyribose.
Nucleosides are components of nucleotides. A nucleotide is the monomeric unit of nucleic acid, e.g. DNA and RNA. In two-stranded nucleic acids like DNA, the nucleobases are paired. The two nucleobases that are complementary are connected by a hydrogen bond.
Etymology: nucleic- (“nucleus“) + -ose + -ide
Characteristics of Nucleoside
A nucleoside is a purine or a pyrimidine nucleobase with a pentose sugar component, which is either ribose or deoxyribose. Therefore:
nucleoside = nucleobase + ribose or deoxyribose
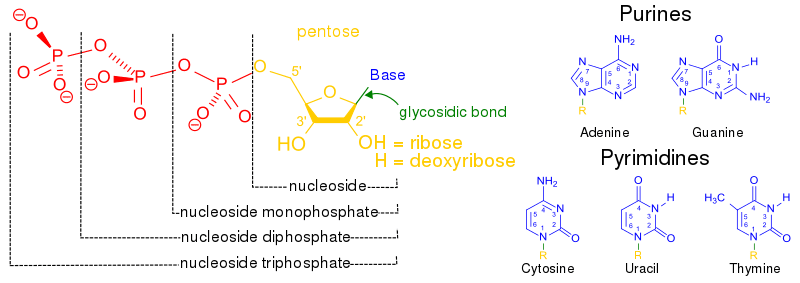
A nucleoside is a glycoside formed from the hydrolysis of nucleic acid. When a phosphate group is covalently attached to the pentose sugar of a nucleoside, it forms a nucleotide. Thus:
nucleotide = nucleoside + phosphate group
In a nucleoside, the anomeric carbon is attached to the N9 of a purine (or to the N1 of a pyrimidine) by a glycosidic bond.
Watch this vid about nucleosides:
Types of Nucleoside
Depending on the pentose sugar component, a nucleoside may be a ribonucleoside or a deoxyribonucleoside.
- A ribonucleoside is a nucleoside with a ribose sugar component. Depending on the nucleobase component, the ribonucleoside may be:
- A deoxyribonucleoside is a nucleoside with deoxyribose sugar. Depending on the nucleobase component, a deoxyribonucleoside may be:
- Deoxyadenosine (nucleobase: adenine)
- Deoxyguanosine (nucleobase: guanine)
- Deoxycytidine (nucleobase: cytidine)
- Thymidine (or deoxyuridine) (nucleobase: uracil)
Also, depending on the nucleobase component, the nucleosides may be grouped into either the “double-ringed” purine or the “single-ringed” pyrimidine.
Examples of Nucleosides
- Adenosine is a purine nucleoside that has adenine bound to a ribose sugar by a glycosidic bond. It is found in all living organisms as a structural component of important biomolecules such as DNA and RNA. It is also a major molecular component of ATP, ADP, and AMP. It is thus involved in various physiologic processes such as energy transfer (e.g. as ATP) and signal transduction (i.e. as cAMP). It also acts as an inhibitory neurotransmitter, presumably in promoting sleep.
- Guanosine is a purine nucleoside that has guanine bound to a ribose sugar. It may be converted into nucleotides: guanosine monophosphate (GMP), cyclic guanosine monophosphate (cGMP), guanosine diphosphate (GDP) or guanosine triphosphate (GTP) through phosphorylation. These nucleotides are involved in various biochemical processes, e.g. nucleic acid synthesis, protein synthesis, photosynthesis, muscle contraction, and intracellular transduction.
- Cytidine is a pyrimidine nucleoside that has cytosine attached to the pentose sugar ribose. It may have an antidepressant effect as it could regulate neuronal-glial glutamate clycling.1
- Uridine is a ribonucleoside that has uracil attached to a ribose ring. It is a white, odorless powder important in carbohydrate metabolism.
- 5-methyluridine is a pyrimidine nucleoside. It contains a thymine nucleobase joined to the ribose sugar. It is a small white crystalline solid.
- Deoxyadenosine is a purine nucleoside that has adenine bound to a deoxyribose sugar. Deoxyadenosine differs from adenosine by having a deoxyribose sugar moiety.
- Deoxyguanosine is a purine nucleoside that has guanine attached to a deoxyribose sugar. Deoxyguanosine differs from guanosine by having deoxyribose as its sugar component instead of ribose.
- Deoxycytidine is a pyrimidine nucleoside that has cytosine attached to a deoxyribose ring. It differs from cytidine with one oxygen atom removed.
- Thymidine is a pyrimidine nucleoside that has thymine bound to a deoxyribose sugar. Thymidine is in fact a deoxyribonucleoside. The prefix _deoxy_– is often left out since there are no precursors of thymine nucleotides involved in RNA synthesis, only DNA synthesis. Hence, it is sometimes simply called thymidine.
- Deoxyuridine is a deoxyribonucleoside that has uracil attached to a deoxyribose ring. It is an antimetabolite, especially when it is converted to deoxyuridine triphosphate and incorporated to DNA during replication. The presence of side groups bound to the uracil component prevents base pairing, hence, interferes with DNA synthesis.
- Inosine is another nucleoside. One of the ways by which it forms is when hypoxanthine is attached to a ribose ring via a β-N9-glycosidic bond. Inosine can be found typically in tRNAs. Inosine is also involved in purine nucleotide reactions during muscle movements.
Nucleoside Metabolism
Nucleosides may be produced de novo in the liver. They may also be available in the diet containing nucleic acids. The nucleic acids are digested into nucleotides.
The nucleotides are then broken down into nucleosides by the action of nucleotidases. The nucleosides, in turn, may be broken down in the lumen of the digestive system into their subcomponents (i.e. nucleobase and sugar) through the action of nucleosidases. Intracellular degradation of nucleosides occurs to yield nucleobases and ribose-1-phosphate or deoxyribose-1-phosphate.
Biological Functions of Nucleosides
Nucleosides, when phosphorylated by kinases, form nucleotides, which in turn, serve as the monomeric unit of nucleic acids. Thus, nucleosides are biologically important as they serve as a structural constituent of DNA and RNA. DNA essentially carries the genetic code whereas RNAs, such as mRNAs, tRNAs, and rRNAs, are involved in translating this genetic code into specific proteins.
Apart from nucleic acids, adenosine triphosphate (ATP), which is a type of nucleotide, is an energy-rich molecule. It contains a large amount of chemical energy stored in its high-energy phosphate bonds. It releases energy when it is broken down (hydrolyzed) into adenosine diphosphate (ADP). The energy is used for many metabolic processes. Hence, ATP is considered the universal energy currency for metabolism.
Medical Importance
Nucleoside analogs are produced artificially for use as therapeutic drugs. They have antiviral properties and therefore are used to prevent further growth of pathogenic viruses inside the host cell. They may also be used as anticancer agents.
Take the Nucleoside Biology Quiz!
Reference
- Machado-Vieira, R., Salvadore, G., Diaz Granados, N., Ibrahim, L., Latov, D., Wheeler-Castillo, C., Baumann, J., Henter, I. D., & Zarate, C. A. (2010). “New Therapeutic Targets for Mood Disorders”. The Scientific World Journal 10: 713–726. doi:10.1100/tsw.2010.65
© Biology Online. Content provided and moderated by Biology Online Editors