Association and mechanism of garlic consumption with gastrointestinal cancer risk: A systematic review and meta-analysis (original) (raw)
Abstract
Gastrointestinal cancer is one of the most commonly diagnosed cancer type worldwide, with millions of cases per year. The aim of this review was to investigate the relationship between garlic intake and the risk reduction of gastrointestinal cancer. We performed saturated data mining on various public domain databases, including PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase (https://www.embase.com/landing?status=grey), and Cochrane Library (https://www.cochranelibrary.com/), with key terms including: ‘garlic’, ‘allium’, ‘stomach’, ‘gastric’, ‘colon’, ‘neoplasms’, ‘cancer’ and ‘tumor’. Furthermore, we identified additional references through expert manual curation. Finally, a meta-analysis was conducted to determine whether garlic intake reduces the risk of gastric and/or colorectal cancer. The association between garlic intake and reduction in the risk of gastric cancer [odds ratio (OR)=0.65, 95% confidence interval (CI)=0.49-0.87, P<0.001] were clear. Nine studies on garlic intake and colorectal cancer showed that garlic reduced cancer risk with a statistical significance (OR=0.75, 95% CI=0.65-0.87, P<0.001). We summarized that four main organic sulfides in garlic, diallyl disulfide (DADS), diallyl trisulfide (DATS), S-allylmercaptocysteine (SAMC) and allicin, may contribute to the regulation of tumor cell apoptosis, migration and the cell cycle. We identified the association between garlic intake and reduced risk of gastric and colorectal cancers and hypothesized that the active ingredients in garlic may act on multiple pathways to reduce the risk of gastrointestinal tumors according to published papers. Importantly, the potential tumor-preventing effect of these garlic ingredients warrants further investigation in regards to the specific mechanism of the underlying antitumor activities.
Keywords: gastrointestinal cancer, garlic, gastric cancer, meta-analysis
Introduction
Gastrointestinal cancer is a health issue with worldwide concern, of which gastric and colorectal cancers are the most common types (1,2). Despite the declining incidence and mortality, gastric cancer remains the third leading cause of cancer-related mortality in the world (1,3). Nearly one million gastric cancer cases are diagnosed worldwide yearly, about half of which are found in the Chinese population (3). The incidence of colorectal cancer ranks third in the world, with highest morbidity and mortality in Asian populations (4). This distribution may be related to particular diet habits, increased level of stress and/or the Helicobacter pylori (H. pylori) infection prevalence in the Asian population (5,6). China and South Korea prefer high-salt foods such as pickles and kimchi. Koreans consume more than twice the daily salt intake recommended by the World Health Organization (7,8), and a high-salt diet can lead to a series of gastrointestinal diseases. Approximately half of the world's population is infected with H. pylori, while more than 55% are found in China (9,10). Some studies have shown the relationship among vegetable consumption, gastrointestinal tumors and H. pylori (11–14), confirming that the increased consumption of fibers that are abundant in fresh fruits and vegetables is correlated with a reduced risk of gastrointestinal cancer (11).
Historically, garlic consumption has been associated with medicinal properties in ancient cultures of Indochina, the Mediterranean and Northern Africa (15). Garlic was shown to be able to reduce the risk of carcinogenesis in breast cancer, pancreatic cancer and esophageal cancer models (16–18). The S-allyl cysteine, diallyl disulfide, and other compounds found in garlic were suggested to have anticancer effects in cellular models (15,19,20). Many potential anticancer mechanisms of these compounds were proposed, including the inhibition of cell proliferation, changes in enzyme activity and immune regulation (21,22). The active ingredients in garlic oil correspond mainly to a family of organosulfur molecules, which selectively increase redox stress in cancer cells, leading to apoptosis and death (23).
Previous meta-analyses and reviews exploring the relationship between garlic consumption and the risk of gastric and colorectal cancers have come to inconsistent conclusion (4,14,24–30). While some studies have found that garlic intake could reduce the risk of gastric and colorectal cancers (14,30), others have shown that this effect may be overestimated (28). In a recent study by Li et al (13) with a follow-up of 22.3 years, garlic supplementation was found to be associated with reduced gastric cancer mortality (OR=0.81, 95% CI=0.57-1.13), with a delayed effect on gastric cancer mortality. Although this finding provides a potential opportunity for the prevention of gastric cancer, further large-scale intervention trials are needed to confirm the effect. Based on the prospective data from the Nurses' Health Study involving 121,700 nurses [relative risk (RR)=1.21, 95% CI=0.94-1.57] and the Health Professionals Follow-up Study (RR=1.00, 95% CI=0.71-1.42) involving 512,529 male health professionals, Meng et al (31) found no association between garlic consumption and the risk of colorectal cancer. However, this study was excluded in this research due to the lack of OR or RR data. Different diets in various populations, various levels of garlic consumption, and diverse patterns of garlic intake may cause inconsistent results from the different studies. Therefore, the effect of garlic on gastrointestinal cancer needs to be further confirmed. We conducted this meta-analysis to update the epidemiological evidence for the association between garlic and gastrointestinal cancer.
Materials and methods
Search strategy
This systemic review and meta-analysis is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. The study was registered in PROSPERO (CRD42020179464). The authors completed the data search in September 2021. All relevant studies that related to garlic intake for gastric and colorectal cancers from 1980 to 2021 were identified by searching in the following databases: Pubmed (https://pubmed.ncbi.nlm.nih.gov/), Embase (https://www.embase.com/landing?status=grey) and Cochrane Library (https://www.cochranelibrary.com/), with key terms including: ‘garlic’, ‘allium’, ‘stomach’, ‘gastric’, ‘colon’, ‘neoplasms’, ‘cancer’ and ‘tumor’. The detailed searching strategies in each database are shown in Tables SI-SIII. All studies that met the requirements were reviewed. In addition, we expanded the search fields by including the references of the screened articles.
Study selection
During the database compilation, two investigators (YaW and DL) reviewed the full text of all the screened publications to determine whether the studies met the selection criteria. Further refinement of the database was completed by a third investigator (YuW). Studies were selected according to the following criteria: i) randomized controlled trials, case-control trials, or with cohort design; ii) studies that include the evaluation of the association between garlic intake and gastric or colorectal cancers over nearly 30 years; iii) studies that provide odds ratio (OR) or relative risk (RR) and with 95% confidence interval (CI) or providing sufficient information for OR/RR and 95% CI calculation; iv) studies published within the last 30 years. The exclusion criteria included: i) reviews or meta-analyses; ii) non-English literature; iii) studies that lacked OR or RR data, or without sufficient data estimation results; iv) studies for which animal, cell, in vitro, and in vivo experiments were excluded. Since the majority of related studies were published in English, we chose not to include non-English studies which were very few and had lack of representativeness. The studies that were included were all non-truncated ones.
Data extraction
Data mining was performed by two investigators. Disagreements were resolved by consultation with a third investigator. The following information was extracted: author, year of publication, study period, study type, country, number of subjects, risk estimates and their 95% CI, description of garlic intake categories, and adjusted variables.
Risk of bias assessment
For randomized controlled trials (RCTs), we assessed the risk of bias using the Cochrane Risk of Bias assessment tool (32). The following characteristics were evaluated: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other biases. According to the recommendations of the Cochrane Handbook, a judgment to risk of bias was determined as three categories, including low risk, unclear risk and high risk. We used the Newcastle-Ottawa Scale (NOS) to assess the risk of bias in nonrandomized studies and scored the studies in three categories: selection (four questions), comparability of study groups (two questions), and ascertainment of exposure or outcome (three questions). Regarding the comparability, the study groups were awarded a maximum of two points; all the other questions were assigned a score of one point (33).
Statistical analysis
We first collected the OR of gastric cancer in various studies. Since the incidences of gastric cancer and colorectal cancer are relatively low, the approximate OR was obtained based on the RR. Then we explored the sources of heterogeneity and conducted a subgroup analysis by garlic intake level, geographic area, and the type of study.
The heterogeneity was assessed using the Cochrane's Q test and I2 statistic. P-values <0.1 and I2 values >50% suggested the existence of heterogeneity. If significant heterogeneity existed, a random effect model was selected; otherwise, the fixed-effects model was used. Meanwhile, I2 values of <30%, 30–60%, and >60% were considered to indicate low, moderate, and high heterogeneity, respectively. Results were assessed using forest plots. All data analysis was performed by STATA 12.0 (https://www.stata.com/).
Sensitivity analysis and subgroup analysis
Sensitivity analysis was performed to identify potential sources of heterogeneity according to garlic consumption level, research type and geographical area. Subgroup analysis was conducted to identify the cause of heterogeneity. Random effect model and fixed effect model were selected according to different degrees of heterogeneity.
Publication bias
Publication bias was assessed by conducting Begg's and Egger's funnel plot asymmetry tests, a P-value <0.1 suggested publication bias with statistical significance.
Results
Study selection and characteristics
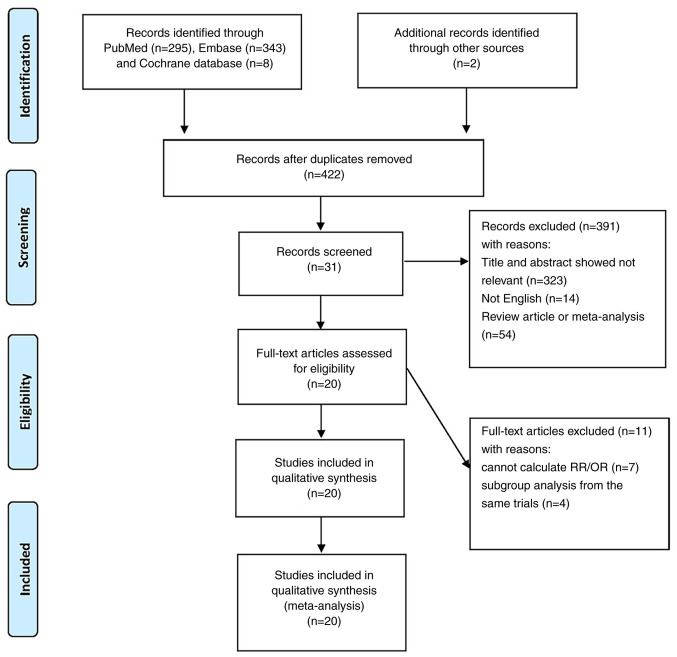
A total of 648 articles were initially identified, of which 226 articles were excluded as duplicate studies. Then we reviewed the titles and abstracts of each literature study according to inclusion and exclusion criteria. We excluded additional articles, among which 323 were irrelevant to this study, 54 were meta-analyses and review, and 14 were non-English literature. After a careful review of full texts in the remaining 31 articles, we finally included 20 articles after excluding 4 articles from the same study and 7 articles with insufficient data (12,13,18,34–50). The flow of the literature search is documented in Fig. 1.
Figure 1.
Flow diagram of the included studies. A total of 648 articles were initially searched, of which 226 articles were excluded as duplicate studies. Then we reviewed the titles and abstracts of each literature according to inclusion and exclusion criteria. We excluded additional articles, among which 323 were irrelevant to this study, 54 were meta-analyses and reviews, and 14 were non-English literature. After a careful review of the full texts in the remaining 31 articles, we finally included 20 articles after excluding 4 articles from the same study and 7 articles with insufficient data. OR, odds ratio; RR, relative risk.
Of the 20 included articles (Table I), 11 were about garlic and gastric cancer, and 9 were about garlic and colorectal cancer. The 11 studies on garlic and gastric cancer were published between 1989 and 2020, including 3,299 patients with gastric cancer and 133,801 controls from one randomized controlled trial (13), 8 case-control studies (18,34,35,37,38,40,41,47) and two cohort studies (36,39). The study by Setiawan et al (35) was a large population-based case-control study of Shanghai and Qingdao, thus we divided this study into a and b to represent the results of Shanghai and Qingdao, respectively. The study by Kim et al (39) was composed of The Nurses' Health Study (NHS) and The Health Professionals Follow-up Study (HPFS); therefore, we split the results into two parts. The 9 studies on garlic and colorectal cancer include 7 case-control studies (12,43,44,46,48–50) and two cohort studies (42,45), published between 1994 and 2018 involving 8,519 colorectal cancer patients and 52,423 controls. Of the 9 studies, 4 were conducted in Europe, 2 in Asia, 2 in the US and 1 in Australia. Both Franceschi et al (46) and Dorant et al (42) studies included colon and rectal cancer, thus we believe that it was reasonable to separate colon and rectal cancers.
Table I.
Details of all 20 studies included in this analysis.
| Authors of the study (year) | Country | Study design | Study period | Cases/Controls | Garlic consumption types | Consumption | OR/RR (95% CI) | Adjustment | (Refs.) |
|---|---|---|---|---|---|---|---|---|---|
| You et al (1989) | China | Population-based case-control study | 1984- 1986 | 564/1,131 | Garlic | 0 (kg/year) 0.1-1.5 (kg/year) >1.5 (kg/year) | 1.00 0.8 (0.5-1.2) 0.7 (0.4-1.0) | Age, sex, family income, and intake of other allium vegetables | (38) |
| Dorant et al (1996) | The Netherlands | Cohort study | 1986-1990 | 152 (male 119, female 33)/3,340 (male 1,627, female 1,713) | Garlic supplement | No supplements Exclusively garlic | 1.00 1.27 (0.61-2.64) | Age, alcohol intake, vitamin C intake, and b-carotene as continuous variables and sex, smoking status, education, history of stomach di-orders, and family history of stomach cancer as categorical variables | (36) |
| Gao et al (1999) | China | Population-based case-control study | 1995-1997 | 153 (male 140, female 53)/234 (male 154, female 80) | Garlic | <1 time/month 1–3 times/month ≥1 time/week | 1.00 0.4 (0.21-0.76) 0.31 (0.22-0.44) | Age, sex, income, smoking, drinking, tea consumption and intake of leftover gruel, pickled vegetables, meat, fruit, tomatoes, eggs and snap beans. | (40) |
| Muñoz et al (2001) | Venezuela | Population- based case-control study | 1991-1997 | 292/485 | Garlic | Less than once/week Several times/week Every day | 1.00 0.7 (0.4-1.0) 0.5 (0.3-0.8) | Age, sex and SES | (37) |
| Takezaki et al (2001) | China | Population-based case-control study | 1996-2000 | 187 (male 137, female 50)/333 (male 235,) female 98 | Garlic | <1 time/week 1–2 times/week 3–5 times/week Everyday | 1.00 1.00 (0.56-1.81) 0.72 (0.42-1.24) 0.66 (0.37-1.17) | Age, sex, smoking and drinking habits | (18) |
| De Stefani et al (2001) | Uruguay | Hospital-based case-control study | 1997-2000 | 160 (male 114, female 46)/320 (male 225, female 95) | Garlic | 0.67 (0.38-1.18) | Age, sex, residence, urban/rural status, education, body mass index (BMI), and total energy intake, and total fruit intake | (34) | |
| Setiawan et al (2005a) | China | Population-based case-control study | 1991-1993 | 750 (male 478, female 272)/750 (male 478, female 272) | Garlic | Never Occasional Often | 1.00 1.11 (0.87-1.41) 0.68 (0.37-1.26) | Matching variables (age, sex), education, BMI, pack-years of smoking, alcohol drinking, salt intake, and vegetable and fruit intake | (35) |
| Setiawan et al (2005b) | China | Population-based case-control study | 1991-1993 | 201 (male 143, female 58)/201 (male143, female 58) | Garlic | Never Occasional Often | 1.00 0.71 (0.27-1.88) 0.45 (0.15-1.30) | Matching variables (age, sex), education, BMI, pack-years of smoking, alcohol drinking, salt intake, and vegetable and fruit intake | (35) |
| Pourfarzi et al (2009) | Iran | Population-based case-control study | 1999- 2005 | 217 (male 151, female 66)/394 (male 265, female 129) | Garlic | Never or infrequently 1–2 times/week >3 times/week | 1.00 0.48 (0.25-0.91) 0.35 (0.13-0.95) | Sex, age group, education, family history of GC, citrus fruits, garlic, onion, red meat, fish, dairy products, strength and warmth of tea, preference for salt intake and H. pylori. | (41) |
| Kim et al (2018) NHS | USA | Cohort study | 1984-2014 | 138/76,948 | Garlic | Never 0-<1 per week 1–4 per week 5 per week | 1.00 1.08 (0.67-1.73) 0.81 (0.50-1.31) 1.34 (0.72-2.47) | Age, Caucasian, BMI, physical activity, smoking status, alcohol consumption, current multivitamin use, current aspirin use, personal history of diabetes mellitus, and intakes of total calorie, red/processed meat, fruits, vegetables, and coffee | (39) |
| Kim et al (2018) HPFS | USA | Cohort study | 1984-2014 | 154/46,244 | Garlic | Never 0-<1 per week 1–4 per week 5 per week | 1.00 1.13 (0.75-1.72) 1.15 (0.74-1.77) 1.45 (0.76-2.78) | Age, Caucasian, BMI, physical activity, smoking status, alcohol consumption, current multivitamin use, current aspirin use, personal history of diabetes mellitus, and intakes of total calorie, red/processed meat, fruits, vegetables, and coffee | (39) |
| Li et al (2019) | China | Randomized controlled trial | 1995-2017 | 151/3,241 | Garlic supplementation | Twice a day | 0.81 (0.57-1.13) | Baseline histology, age, sex, history of ever using alcohol, and history of ever smoking | (13) |
| Yuan et al (2020) | China | Hospital-based case-control study | 2014-2016 | 180/180 | Garlic | 0.35 (0.18-0.67) | Dietary/lifestyle habits, psychological factors, serum PG I level, serum PG II level, PG I/II ratio, serum G-17 level and H. pylori infection | (47) | |
| Steinmetz et al (1994) | USA | Prospective cohort Study | 1986-1990 | 212/35004 | Garlic | L1 (0) L2 (0.5) L3 (1.0) | 1.00 1.07 (0.77-1.50) 0.68 (0.46-1.02) | Age and energy intake | (45) |
| Witte et al (1996) | USA | Case control study | 1991-1993 | 488 (male 325, female 163)/488 (male 325, female 163) | Garlic | None 0.5 1.0-2.5 >-3.0 | 1.00 0.92 (0.64-1.34) 0.98 (0.61-1.56) 0.66 (0.43-1.01) | Race, BMI, physical activity, smoking, calories, and saturated fat using conditional logistic, regression dietary fiber, folate, β-carotene, and vitamin C | (49) |
| Dorant et al (1996) (colon) | The Netherlands | Cohort study | 1986-1989 | 293 (male 150, female 143)/3,123 (male 1,525, female 1598) | Garlic supplement | No supplement Exclusively garlic | 1.00 1.36 (0.79-2.35) | Age, vitamin C and (J-carotene as continuous variables, and sex, smoking status, education, family history of intestinal cancer, previous history of chronic intestinal disease or chole cystectomy as categorical variables | (42) |
| Dorant et al (1996) (rectum) | The Netherlands | Cohort study | 1986-1989 | 150 (male 93, female 57)/3,123 (male 1,525, | Garlic supplement | No supplement Exclusively garlic | 1.00 1.28 (0.63-2.60) | Age, vitamin C and (J-carotene as continuous variables, and sex, smoking female 1,598) status, education, family history of intestinal cancer, previous history of chronic intestinal disease or cholecystectomy as categorical variables | (42) |
| Franceschi et al (1997) (colon) | Italy | Case control study | 1991-1996 | 1,225/5,155 | Cooked garlic | 0.9 (0.8-1.0) | Age, sex, center, year of interview, education, physical activity, alcohol and energy intake | (46) | |
| Franceschi et al (1997) (rectum) | Italy | Case control study | 1991-1996 | 728/5,155 | Cooked garlic | 0.9 (0.8-1.0) | Age, sex, center, year of interview, education, physical activity, alcohol and energy intake | (46) | |
| Levi et al (1999) | Switzerland | Case control study | 1992-1997 | 223 (male 142, female 81)/491 (male 211, female 280) | Garlic | Low Medium High | 1.00 0.51 (0.35-0.74) 0.32 (0.18-0.57) | Age, sex, education, smoking, alcohol, BMI, physical activity and total energy intake | (44) |
| Galeone et al (2006) | Italy | Case control study | 1991-2004 | 2,280 (male 1,318, female 962)/4,765 (male 2,403, female 2,362) | Garlic | None or low Intermediate High | 1 0.88 (0.78, 0.98) 0.74 (0.63,0.86) | Age, sex, study center, education, BMI, energy intake, alcohol consumption, smoking habit, and physical activity | (48) |
| Annema et al (2011) | Australia | Population-based case-control study | 2005-2007 | 834 (male 514, female 320)/939 (male 551, female 388) | Garlic | <0.02 (servings/day) 0.02-<0.14 (servings/day) 0.14-<0.28 (servings/day) 0.28+ (servings/day) | 1.00 0.92 (0.67-1.26) 0.84 (0.62-1.15) 0.86 (0.68-1.09) | Adjusted for sex, age, BMI at age 20 years, energy intake, multivitamin use, alcohol consumption, physical activity, smoking, diabetes and socioeconomic status | (50) |
| Wang et al (2018) | China | Case control study | 2015-2016 | 317 (male 145, female 172)/317 (male 146, female 171) | Garlic | 0.499 (0.341-0.732) | (43) | ||
| Wu et al (2019) | China | Hospital-based matched case-control study | 2009-2011 | 833/833 | Garlic | <0.60 (kg/year) 0.60-2.60 (kg/year) 2.60-3.65 (kg/year) >3.65 (kg/year) | 1.00 0.49 (0.35-0.66) 0.43 (0.30-0.59) 0.56 (0.39-0.79) | BMI, family history of CRC (first degree), education level, smoking, passive smoking, alcohol, the consumption of red meat, milk, other vegetables, fruit, total energy, fiber, calcium, fat, vitamin C, vitamin D, cholesterol, and folic acid | (12) |
Overall and subgroup analysis of evidence
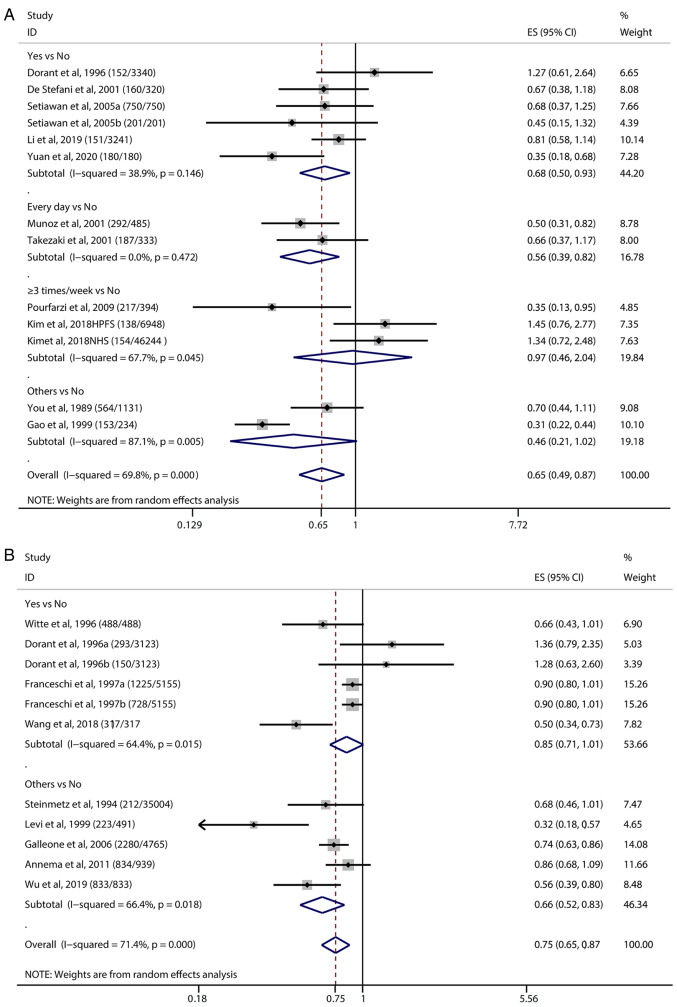
We conducted an overall estimation by categories of garlic consumption (Fig. 2). The ORs of all the studies were extracted for the meta-analysis. The OR obtained by the pooled analysis was 0.65 (95% CI=0.49-0.87, P<0.001), indicating that garlic intake was associated with a lower risk of gastric cancer in individuals compared with those without garlic intake (Fig. 2A). Participants who consumed garlic every day had a significant lower risk of gastric cancer than those who did not consume garlic.
Figure 2.
Associations between garlic intake and gastrointestinal cancer risk by garlic consumption. The (number/number) after each study in the figure indicates the (Cases/Controls). Vertical solid black line: invalid line; red dashed line: pooled effect size; horizontal black solid line: the width of the line represents the confidence interval (CI) of each study, the black diamond in the middle represents the OR of each study, and the gray square represents the weight of each study. Others: We have included some studies that differed from other classifications of garlic intake into this category. (A) Forest plots for the associations between garlic intake and gastric cancer risk by garlic consumption. The OR obtained by the pooled analysis was 0.65 (95% CI=0.49-0.87). (B) Forest plots for the associations between garlic intake and colorectal cancer risk by garlic consumption. The meta-analysis using the random-effects model showed a combined estimated OR of 0.75 (95% CI=0.65-0.87), suggesting that garlic intake could reduce the risk of colorectal cancer. OR, odds ratio; ES, effect size.
In the subgroup analysis by geographic area (Fig. S1), the estimated OR of the studies in Asia, Europe and America was 0.53 (95% CI=0.38-0.73), 1.27 (95% CI=0.61-2.64), 0.87 (95% CI=0.52-1.47, P<0.05), respectively (Table SIV). In addition, the comprehensive analysis of prospective studies showed that garlic intake correlated with a small reduction in gastric cancer (OR=1.07, 95% CI=0.79-1.47), while the retrospective studies showed garlic intake had a more significant effect (OR=0.50, 95% CI=0.39-0.64) (Fig. S2).
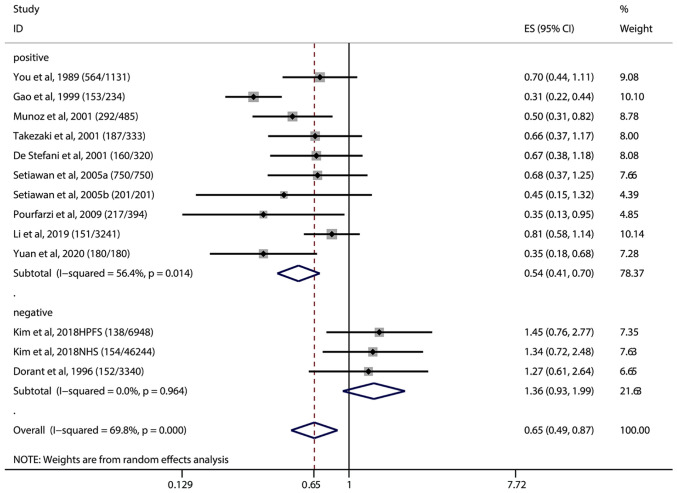
We found that among those 11 included studies, 2 studies (36,39) containing 126,976 subjects showed that garlic intake had no significant association with the incidence of gastric cancer (OR=1.36, 95% CI=0.93-1.99), while 9 studies (13,18,34,35,37,38,40,41,47) containing 9,944 subjects showed that garlic intake could significantly reduce the incidence of gastric cancer (OR=0.54, 95% CI=0.41-0.70) (P<0.05) (Fig. 3).
Figure 3.
Positive and negative association studies between garlic intake and gastric cancer. The (number/number) after each study in the figure indicates the (Cases/Controls), Vertical solid black line: invalid line; red dashed line: pooled effect size; horizontal black solid line: the width of the line represents the confidence interval (CI) of each study, the black diamond in the middle represents the OR of each study, and the gray square represents the weight of each study. Among the 11 included studies, 2 studies showed that garlic intake had no association with the incidence of gastric cancer (OR=1.36, 95% CI=0.93-1.99), including 12,6976 subjects, and 9 studies showed that garlic intake could reduce the incidence of gastric cancer (OR=0.54, 95% CI=0.41-0.70), including 9,944 subjects. OR, odds ratio; ES, effect size.
A total of 9 studies estimated the association between garlic intake and the risk of colorectal cancer (Fig. 2B). The meta-analysis using the random-effects model showed a combined estimated OR of 0.75 (95% CI=0.65-0.87, P<0.001), suggesting that garlic intake could significantly reduce the risk of colorectal cancer. Among the 9 included research studies, only Dorant et al (42) and Franceschsi et al (46) estimated the OR values for colon cancer and rectal cancer separately, without providing the total OR value.
Compared to the retrospective studies (OR=0.72, 95% CI=0.62-0.84, P<0.001), the results of the prospective study (OR=1.01, 95% CI=0.62-1.65, P<0.1) showed an insignificant effect of garlic intake on reducing the risk of colorectal cancer (Table SV; Fig. S3). Subgroup analyses of geographical regions (Fig. S4) showed that garlic intake significantly reduced the risk of colorectal cancer in Asia compare to other regions.
Heterogeneity assessment and sensitivity analysis
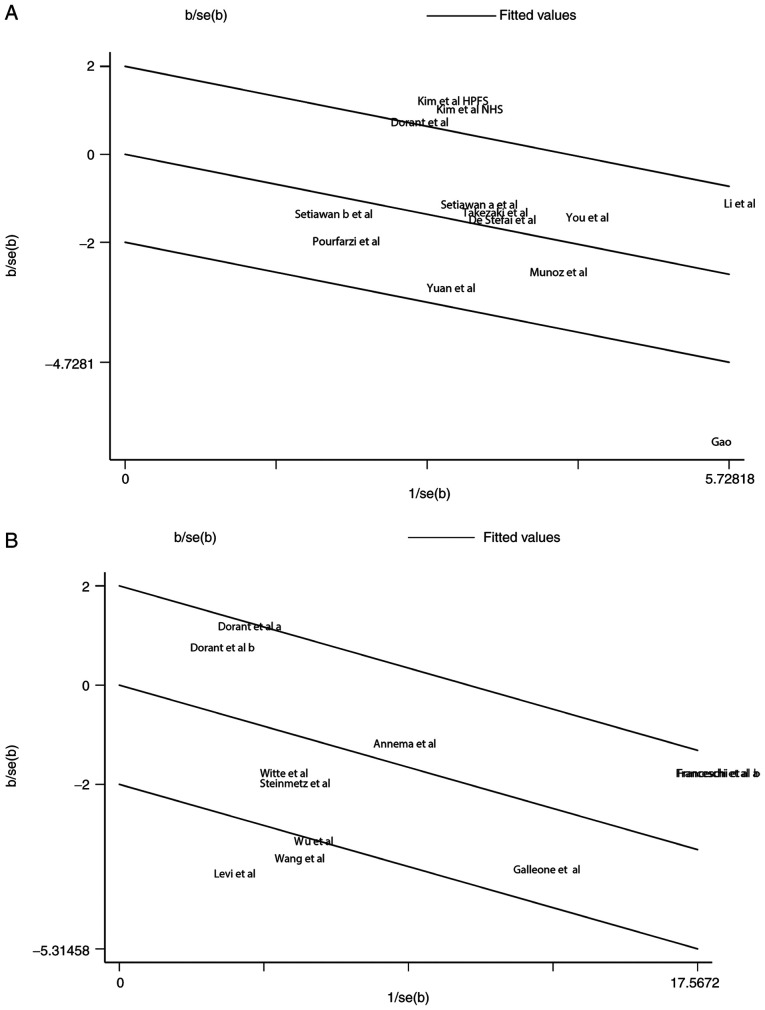
The random effect model suggested a strong heterogeneity with I2=69.8%, and P<0.1 in the studies of garlic and gastric cancer which were selected for the meta-analysis. Therefore, we conducted Galbraith test to further identify the source of heterogeneity. The result of Galbraith test showed that the studies of Gao et al (40) and Kim et al (39) were the main sources of heterogeneity (Fig. 4A). The result of sensitivity analysis showed that our results were stable, and there was no significant difference in the pooled results (Fig. 5A).
Figure 4.
Source of heterogeneity in the meta-analysis of garlic intake and gastrointestinal cancer risk. (A) The Galbraith test indicating the source of heterogeneity in the meta-analysis of garlic intake and gastric cancer risk. The studies of Gao et al (1999) and Kim et al (2018) were the main sources of heterogeneity. (B) The Galbraith test indicating the source of heterogeneity in the meta-analysis of garlic intake and colorectal cancer risk. The results indicate that 3 studies (Wu et al, Levi et al, and Wang et al) were the main sources of heterogeneity.
Figure 5.
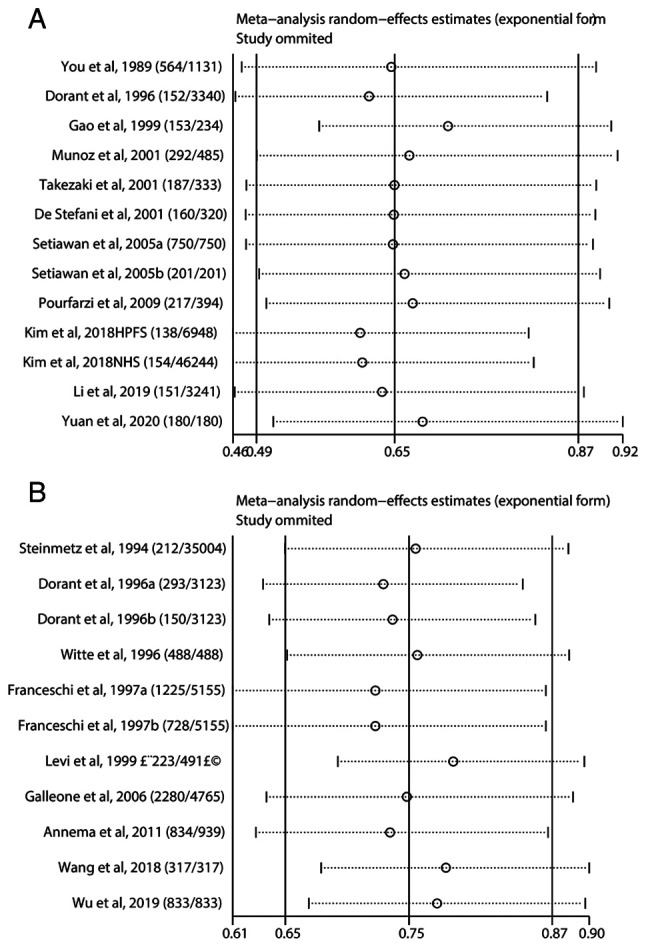
Sensitivity analysis of garlic and gastrointestinal cancer. (A) Sensitivity analysis of garlic and gastric cancer. No significant differences were found among the studies. (B) Sensitivity analysis of garlic intake and colorectal cancer. The results of the sensitivity analysis showed that no articles exceeded the limits and there were no significant differences among the studies.
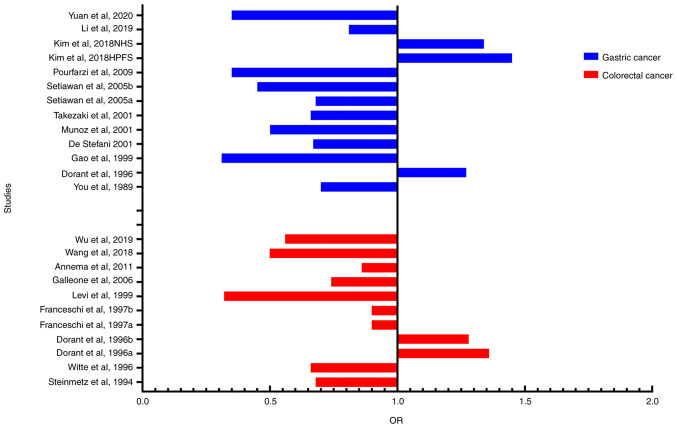
For the 9 studies of garlic and colorectal cancer, a significant heterogeneity was also suggested (I2=71.4%, P<0.001). According to the results of Galbraith test, three studies [Wu _et al_ (12), Levi et al (44) and Wang et al (43)] were indicated as the main sources of heterogeneity (Fig. 4B). Sensitivity analysis was used to estimate the impact of each study on the overall estimate (Fig. 5B). The results of the sensitivity analysis showed that no articles exceeded the limits and there were no significant differences among the studies. Our meta-analysis suggested that garlic can reduce the risk of gastrointestinal cancers, and most of the included studies are consistent with this conclusion (Fig. 6).
Figure 6.
Relationship between included studies and risk of gastrointestinal cancer by garlic intake. In all the included studies, the results of most studies indicated that garlic can reduce the risk of gastrointestinal cancers. OR, odds ratio.
Risk of bias assessment
A randomized controlled trial evaluated by the Cochrane risk assessment tool was rated as low risk, and the non-randomized controlled trials were scored using a NOS scale, as shown in Table SVI and Table SVII.
Publication bias
Potential publication bias was assessed using the Begg (Fig. 7) and Egger tests (Table SVIII). There was no significant evidence of publication bias for gastric and colorectal cancers.
Figure 7.
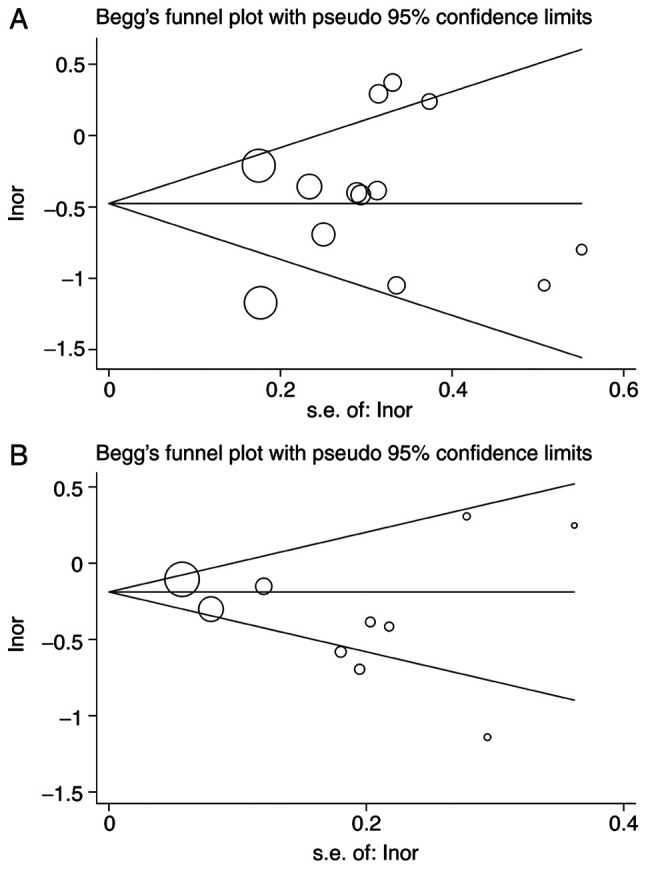
Potential publication bias assessed by Begg's test. Funnel plot indicating the publication bias in the studies included in the meta-analysis of the garlic intake and gastrointestinal cancers. There was no significant evidence of publication bias for gastric and colorectal cancers. (A) Begg's funnel plot for garlic intake and gastric cancer. (B) Begg's funnel plot for garlic intake and colorectal cancer.
Discussion
This meta-analysis combined the results of 20 studies regarding the association of garlic consumption with gastric cancer (11 studies) and colorectal cancer (9 studies). Our results indicated that garlic intake significantly reduces the risk of gastric cancer (OR=0.65, 95% CI=0.49-0.87, P<0.001) and colorectal cancer (OR=0.75, 95% CI=0.65-0.87, P<0.001), consistent with the epidemiological evidence supporting the correlation between garlic intake and a reduced risk of gastric and colorectal cancer. The results of the geographical subgroup analysis showed that a greater risk reduction occurs in the Asian region compared with other geographical regions. We suspect one of the possible reasons is that garlic consumption is higher in Asia, especially in China, where the habit of eating raw garlic leads to a higher consumption than other countries in the world (51,52). Some studies have also analyzed the effects of allium and onion on gastrointestinal tumors. We speculate that the active ingredients may be the same or similar to garlic, and that these foods may have a superimposed effect on gastrointestinal cancers (12,18,38,40,41). In addition, the European population may be under-represented since there was only one study conducted in Europe. Our meta-analysis incorporated the results of the latest research by Li et al (13) and summarized the recent studies. Although most of the included studies were retrospective case-control studies, lacking blinding and randomized control (53), we still found that garlic intake was associated with a reduced risk of gastric and colorectal cancer. Compared to the previous meta-analysis on the relationship between garlic and gastric and colorectal cancer by Fleischauer et al (28), our meta-analysis included more studies and conducted a subgroup analysis with a focus on garlic intake, resulting in more reliable conclusions. Due to the various dietary patterns in the different studies, we cannot exclude the effects of other factors, such as vegetable and fiber intake. Additionally, each study had different confounding factors, and most studies adjusted them, such as sex, age, and others. From the results of the subgroup analysis, it was shown that the intake of garlic can reduce the likelihood of gastric cancer compared with the non-intake of garlic. Although a previous meta-analysis by others showed that the protective effect of garlic on gastric and colorectal cancer may be overestimated (28), the results of the comprehensive analysis in this study indicated the preventive function of garlic in gastrointestinal tumors.
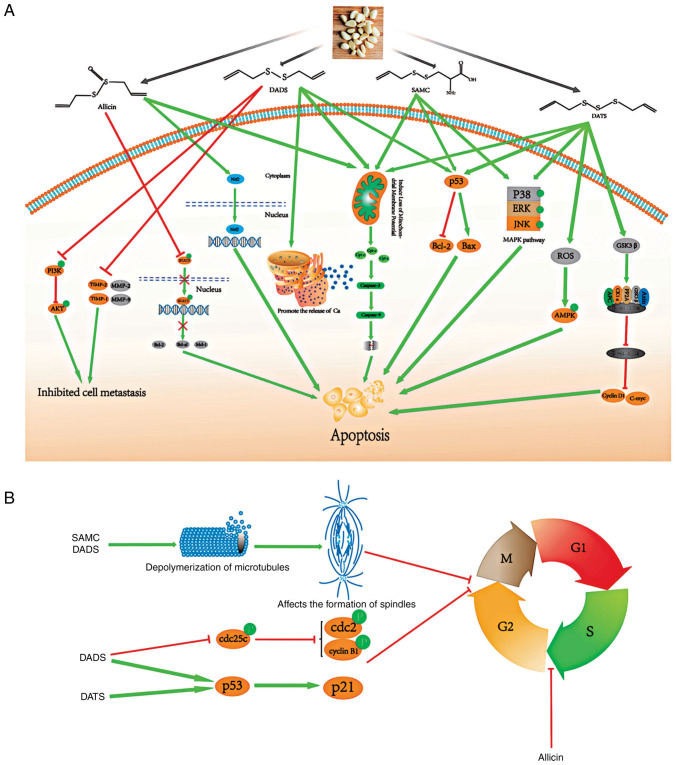
A further review was conducted on the molecular mechanisms of the anticancer effects of garlic (Fig. 8). Based on previous literature, garlic contains a variety of organic sulfur compounds, mainly including S-allylmercaptocysteine (SAMC), diallyl disulfide (DADS), diallyl trisulfide (DATS) and allicin, which are the main components which produce potential antitumor effects. We searched the Pubmed database with key terms including ‘gastrointestinal tumors’, ‘garlic’, ‘mechanism’, ‘pathways’, and reviewed biological functions of these four organic sulfur compounds. These organic sulfur compounds demonstrate potential antitumor activity through various underlying mechanisms. First, organic sulfur compounds can regulate the cell cycle. DADS and DATS can activate the P53/P21 pathway, while DADS can inhibit the expression of cyclin B1, cdc2, and cdc25c proteins, leading to G2/M phase arrest in tumor cells (54,55). SAMC and DADS can inhibit the polymerization of tubulin and thus affect the function of the spindle, resulting in mitotic arrest (56). In addition, allicin induces cell cycle arrest in the S phase of the cell cycle (57). Second, organic sulfides can induce cell apoptosis in the following ways. DADS, DATS, SAMC and allicin promote the release of cytochrome c from mitochondria, thereby activating caspase family proteins, such as caspase 3 and caspase 9, and inducing apoptosis (23,56,58–62); DADS, DATS and SAMC can activate the p53 pathway, resulting in the decreased expression of Bcl-2 and increased expression of Bax (54,58,63–65); SAMC and DATS significantly activate three pathways of the MAPKs pathway, including ERK, JNK and p38 (63,64); Moreover, DATS can significantly upregulate the level of glycogen synthase kinase 3 β (GSK3β) to increase the digestion of β-catenin, indicating that DATS can inhibit the Wnt/β-catenin pathway, a key component in the occurrence and development of tumors. DATS can also increase reactive oxygen species (ROS) production and activate the AMPK pathway (23,55); Allicin can reduce phosphorylated signal transducer and activator of transcription 3 (STAT3) to inhibit the STAT3 pathway, as well as activate Nrf2 and induce its translocation to the nucleus (66,67). Third, DADS can inhibit matrix metalloproteinase (MMP)-2, MMP-9, tissue inhibitor of metalloproteinases-1 (TIMP-1), TIMP-2 and PI3K/AKT pathways to inhibit cell metastasis (68).
Figure 8.
Potential molecular mechanisms of the anticancer effects of garlic. (A) DADS, DATS, SAMC and allicin can all promote the release of cytochrome c from mitochondria, thereby activating the caspase family proteins, such as caspase 3 and caspase 9, and inducing apoptosis; DADS, DATS and SAMC can activate the p53 pathway, resulting in decreased expression of Bcl-2 and increased expression of Bax. SAMC and DATS significantly activated the three pathways of MAPKs, including ERK, JNK and p38. In addition, the Wnt/β-catenin pathway plays a key role in the occurrence and development of tumors, DATS can significantly upregulate the level of GSK3β, thereby increasing the digestion of β-catenin, indicating that DATS can inhibit the Wnt/β-catenin pathway. DATS can also increase ROS production and activate the AMPK pathway. Allicin can reduce phosphorylated STAT3, thereby inhibiting the STAT3 pathway, and allicin can also activates Nrf2 and induces its translocation to the nucleus. Moreover, DADS can inhibit MMP-2 and MMP-9. (B) DADS and DATS can activate the P53/P21 pathway, and DADS can also inhibit the expression of cyclin B1, cdc2, and cdc25c proteins, leading to G2/M phase arrest of tumor cells. SAMC and DADS can affect the polymerization of tubulin and thus affect the function of the spindle, leading to mitotic arrest. Finally, allicin induces cell cycle arrest in the S phase. DADS, diallyl disulfide; DATS, diallyl trisulfide; SAMC, S-allylmercaptocysteine; GSK3β, glycogen synthase kinase 3 β; ROS, reactive oxygen species; STAT3, signal transducer and activator of transcription 3; MMP, matrix metalloproteinase.
To summarize, DADS, DATS, SAMC, and allicin participate in tumor-related biological process through various mechanisms, eventually leading to apoptosis, cell cycle arrest, and migration inhibition in tumor cells. A medical compound containing active ingredients from garlics may exert potential tumor preventive or therapeutic effects through the above-mentioned mechanisms in the human body, representing a novel antitumor treatment alternative.
This meta-analysis has the following limitations. i) Only a small set of randomized controlled trials are included in the date, most of which are case-control and cohort studies. Compared with randomized controlled trials, case-control and cohort studies have more unaccounted parameters in blind control and follow-up, resulting in higher propensity of bias. ii) This meta-analysis included studies conducted in different countries since the 1990s. Not all studies were primarily based on onion vegetables, and there was inconsistent stratification among the studies. iii) Most of the included studies were conducted in China, where the incidence of gastric cancer is generally higher than the rest of the world. Moreover, garlic intake is relatively high in the diet of Chinese people. iv) Many studies did not control other diets, and the type of garlic consumption remains unstandardized. It is difficult to determine the minimum garlic intake for a tumor-protective effect. The minimum and maximum consumption levels varied greatly among the different studies.
The quantified I2 test showed that the included studies had significant heterogeneity, and Galbraith test suggested that some studies might be the sources. Therefore, we explored the possible cause for the heterogeneity. First, most of the included studies were retrospective studies with various confounding factors, and recall bias may have produced different results from the prospective studies. Second, most studies had collected data in the form of questionnaires instead of objective measurement. Third, studies conducted in Asia, especially in China, where garlic is a highly consumed food, may lead to certain bias on the results when pooled together with studies conducted in other places with much lower garlic consumption.
In summary, our meta-analysis provides strong evidence that garlic can reduce the risk of gastric and colorectal cancers. The conclusion was mainly based on case-control studies with many potential confounders, and further research is warranted to validate it.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding Statement
The present work was financially supported by grants from the National Natural Science Foundation of China [32000098, 31671468], the China Postdoctoral Science Foundation [2020M682167, 2020T130070ZX], the Science and Technology Development Plan of Jinan Municipal Health Commission [2020-3-09], Jinan 2020 Science and Technology Innovation Development Plan [202019035], and the Academic Promotion Programme of Shandong First Medical University [2019QL024].
Availability of data and materials
The study was registered in PROSPERO (CRD42020179464).
Authors' contributions
HJL designed the review and meta-analysis. YYW and HJL conceived and wrote the review. YFW and DRL acquired and analysed the data. MYJ and PH analyzed and confirmed the integrity of the data found in the literature. YSW was involved in drafting the manuscript. All authors contributed to the analysis, reviewed the results and read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Authors' information
ORCID: Huanjie Li, orcid.org/0000-0002-4997-0927; Yunshan Wang, orcid.org/0000-0003-3767-6728.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 2.Gausman V, Dornblaser D, Anand S, Hayes RB, O'Connell K, Du M, Liang PS. Risk factors associated with early-onset colorectal cancer. Clin Gastroenterol Hepatol. 2020;18:2752–2759.e2. doi: 10.1016/j.cgh.2019.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 4.Zhou X, Qian H, Zhang D, Zeng L. Garlic intake and the risk of colorectal cancer: A meta-analysis. Medicine (Baltimore) 2020;99:e18575. doi: 10.1097/MD.0000000000018575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Larsson SC, Orsini N, Wolk A. Processed meat consumption and stomach cancer risk: A meta-analysis. J Natl Cancer Inst. 2006;98:1078–1087. doi: 10.1093/jnci/djj301. [DOI] [PubMed] [Google Scholar]
- 6.Sugano K. Effect of Helicobacter pylori eradication on the incidence of gastric cancer: A systematic review and meta-analysis. Gastric Cancer. 2019;22:435–445. doi: 10.1007/s10120-018-0876-0. [DOI] [PubMed] [Google Scholar]
- 7.Park HK, Lee Y, Kang BW, Kwon KI, Kim JW, Kwon OS, Cobb LK, Campbell NRC, Blakeman DE, Kim CI. Progress on sodium reduction in South Korea. BMJ Glob Health. 2020;5:e002028. doi: 10.1136/bmjgh-2019-002028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee HS, Duffey KJ, Popkin BM. Sodium and potassium intake patterns and trends in South Korea. J Hum Hypertens. 2013;27:298–303. doi: 10.1038/jhh.2012.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ford AC, Yuan Y, Moayyedi P. Helicobacter pylori eradication therapy to prevent gastric cancer: Systematic review and meta-analysis. Gut. 2020;69:2113–2121. doi: 10.1136/gutjnl-2020-320839. [DOI] [PubMed] [Google Scholar]
- 10.Hooi JKY, Lai WY, Ng WK, Suen MMY, Underwood FE, Tanyingoh D, Malfertheiner P, Graham DY, Wong VWS, Wu JCY, et al. Global prevalence of Helicobacter pylori infection: Systematic review and meta-Analysis. Gastroenterology. 2017;153:420–429. doi: 10.1053/j.gastro.2017.04.022. [DOI] [PubMed] [Google Scholar]
- 11.Hou N, Huo D, Dignam JJ. Prevention of colorectal cancer and dietary management. Chin Clin Oncol. 2013;2:13. doi: 10.3978/j.issn.2304-3865.2013.04.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu X, Shi J, Fang WX, Guo XY, Zhang LY, Liu YP, Li Z. Allium vegetables are associated with reduced risk of colorectal cancer: A hospital-based matched case-control study in China. Asia Pac J Clin Oncol. 2019;15:e132–e141. doi: 10.1111/ajco.13133. [DOI] [PubMed] [Google Scholar]
- 13.Li WQ, Zhang JY, Ma JL, Li ZX, Zhang L, Zhang Y, Guo Y, Zhou T, Li JY, Shen L, et al. Effects of Helicobacter pylori treatment and vitamin and garlic supplementation on gastric cancer incidence and mortality: Follow-up of a randomized intervention trial. BMJ. 2019;366:l5016. doi: 10.1136/bmj.l5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li Z, Ying X, Shan F, Ji J. The association of garlic with helicobacter pylori infection and gastric cancer risk: A systematic review and meta-analysis. Helicobacter. 2018;23:e12532. doi: 10.1111/hel.12532. [DOI] [PubMed] [Google Scholar]
- 15.Nicastro HL, Ross SA, Milner JA. Garlic and onions: Their cancer prevention properties. Cancer Prev Res (Phila) 2015;8:181–189. doi: 10.1158/1940-6207.CAPR-14-0172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Desai G, Schelske-Santos M, Nazario CM, Rosario-Rosado RV, Mansilla-Rivera I, Ramírez-Marrero F, Nie J, Myneni AA, Zhang ZF, Freudenheim JL, Mu L. Onion and garlic intake and breast cancer, a case-control study in Puerto Rico. Nutr Cancer. 2020;72:791–800. doi: 10.1080/01635581.2019.1651349. [DOI] [PubMed] [Google Scholar]
- 17.Chan JM, Wang F, Holly EA. Vegetable and fruit intake and pancreatic cancer in a population-based case-control study in the San Francisco bay area. Cancer Epidemiol Biomarkers Prev. 2005;14:2093–2097. doi: 10.1158/1055-9965.EPI-05-0226. [DOI] [PubMed] [Google Scholar]
- 18.Takezaki T, Gao CM, Wu JZ, Ding JH, Liu YT, Zhang Y, Li SP, Su P, Liu TK, Tajima K. Dietary protective and risk factors for esophageal and stomach cancers in a low-epidemic area for stomach cancer in Jiangsu Province, China: Comparison with those in a high-epidemic area. Jpn J Cancer Res. 2001;92:1157–1165. doi: 10.1111/j.1349-7006.2001.tb02135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.You WC, Chang YS, Heinrich J, Ma JL, Liu WD, Zhang L, Brown LM, Yang CS, Gail MH, Fraumeni JF, Jr, Xu GW. An intervention trial to inhibit the progression of precancerous gastric lesions: Compliance, serum micronutrients and S-allyl cysteine levels, and toxicity. Eur J Cancer Prev. 2001;10:257–263. doi: 10.1097/00008469-200106000-00009. [DOI] [PubMed] [Google Scholar]
- 20.Wang HR, Pan YM, Zhang L. Mechanism for allicin to sensitize gastric cancer cells to chemotherapy. World Chinese J Digestol. 2019;27:1248–1255. doi: 10.11569/wcjd.v27.i20.1248. [DOI] [Google Scholar]
- 21.Wu S, Li Z. Effect of allicin on the proliferation and apoptosis of gastric cancer cells SGC-7901 through inhibiting autophagy of endoplasmic reticulum. Anti-Tumor Pharmacy. 2019;9:226–230. [Google Scholar]
- 22.Arreola R, Quintero-Fabián S, López-Roa RI, Flores-Gutiérrez EO, Reyes-Grajeda JP, Carrera-Quintanar L, Ortuño-Sahagún D. Immunomodulation and anti-inflammatory effects of garlic compounds. J Immunol Res. 2015;2015:401630. doi: 10.1155/2015/401630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yu CS, Huang AC, Lai KC, Huang YP, Lin MW, Yang JS, Chung JG. Diallyl trisulfide induces apoptosis in human primary colorectal cancer cells. Oncol Rep. 2012;28:949–954. doi: 10.3892/or.2012.1882. [DOI] [PubMed] [Google Scholar]
- 24.Zhu B, Zou L, Qi L, Zhong R, Miao X. Allium vegetables and garlic supplements do not reduce risk of colorectal cancer, based on meta-analysis of prospective studies. Clin Gastroenterol Hepatol. 1991;2014;12 doi: 10.1016/j.cgh.2014.03.019. 2001.e1-4; quiz e121. [DOI] [PubMed] [Google Scholar]
- 25.Turati F, Guercio V, Pelucchi C, La Vecchia C, Galeone C. Colorectal cancer and adenomatous polyps in relation to allium vegetables intake: A meta-analysis of observational studies. Mol Nutr Food Res. 2014;58:1907–1914. doi: 10.1002/mnfr.201400169. [DOI] [PubMed] [Google Scholar]
- 26.Kodali RT, Eslick GD. Meta-analysis: Does garlic intake reduce risk of gastric cancer? Nutr Cancer. 2015;67:1–11. doi: 10.1080/01635581.2015.967873. [DOI] [PubMed] [Google Scholar]
- 27.Hu JY, Hu YW, Zhou JJ, Zhang MW, Li D, Zheng S. Consumption of garlic and risk of colorectal cancer: An updated meta-analysis of prospective studies. World J Gastroenterol. 2014;20:15413–15422. doi: 10.3748/wjg.v20.i41.15413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fleischauer AT, Poole C, Arab L. Garlic consumption and cancer prevention: Meta-analyses of colorectal and stomach cancers. Am J Clin Nutr. 2000;72:1047–1052. doi: 10.1093/ajcn/72.4.1047. [DOI] [PubMed] [Google Scholar]
- 29.Chiavarini M, Minelli L, Fabiani R. Garlic consumption and colorectal cancer risk in man: A systematic review and meta-analysis. Public Health Nutr. 2016;19:308–317. doi: 10.1017/S1368980015001263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Turati F, Pelucchi C, Guercio V, La Vecchia C, Galeone C. Allium vegetable intake and gastric cancer: A case-control study and meta-analysis. Mol Nutr Food Res. 2015;59:171–179. doi: 10.1002/mnfr.201400496. [DOI] [PubMed] [Google Scholar]
- 31.Meng S, Zhang X, Giovannucci EL, Ma J, Fuchs CS, Cho E. No association between garlic intake and risk of colorectal cancer. Cancer Epidemiol. 2013;37:152–155. doi: 10.1016/j.canep.2012.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JAC, et al. The cochrane collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–605. doi: 10.1007/s10654-010-9491-z. [DOI] [PubMed] [Google Scholar]
- 34.De Stefani E, Correa P, Boffetta P, Ronco A, Brennan P, Deneo-Pellegrini H, Mendilaharsu M. Plant foods and risk of gastric cancer: A case-control study in Uruguay. Eur J Cancer Prev. 2001;10:357–364. doi: 10.1097/00008469-200108000-00009. [DOI] [PubMed] [Google Scholar]
- 35.Setiawan VW, Yu GP, Lu QY, Lu ML, Yu SZ, Mu L, Zhang JG, Kurtz RC, Cai L, Hsieh CC, Zhang ZF. Allium vegetables and stomach cancer risk in China. Asian Pac J Cancer Prev. 2005;6:387–395. [PMC free article] [PubMed] [Google Scholar]
- 36.Dorant E, van den Brandt PA, Goldbohm RA, Sturmans F. Consumption of onions and a reduced risk of stomach carcinoma. Gastroenterology. 1996;110:12–20. doi: 10.1053/gast.1996.v110.pm8536847. [DOI] [PubMed] [Google Scholar]
- 37.Muñoz N, Plummer M, Vivas J, Moreno V, De Sanjosé S, Lopez G, Oliver W. A case-control study of gastric cancer in Venezuela. Int J Cancer. 2001;93:417–423. doi: 10.1002/ijc.1333. [DOI] [PubMed] [Google Scholar]
- 38.You WC, Blot WJ, Chang YS, Ershow A, Yang ZT, An A, Henderson BE, Fraumeni JF, Jr, Wang TG. Allium vegetables and reduced risk of stomach cancer. J Natl Cancer Inst. 1989;81:162–164. doi: 10.1093/jnci/81.2.162. [DOI] [PubMed] [Google Scholar]
- 39.Kim H, Keum N, Giovannucci EL, Fuchs CS, Bao Y. Garlic intake and gastric cancer risk: Results from two large prospective US cohort studies. Int J Cancer. 2018;143:1047–1053. doi: 10.1002/ijc.31396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gao CM, Takezaki T, Ding JH, Li MS, Tajima K. Protective effect of allium vegetables against both esophageal and stomach cancer: A simultaneous case-referent study of a high-epidemic area in Jiangsu Province, China. Jpn J Cancer Res. 1999;90:614–621. doi: 10.1111/j.1349-7006.1999.tb00791.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pourfarzi F, Whelan A, Kaldor J, Malekzadeh R. The role of diet and other environmental factors in the causation of gastric cancer in Iran-A population based study. Int J Cancer. 2009;125:1953–1960. doi: 10.1002/ijc.24499. [DOI] [PubMed] [Google Scholar]
- 42.Dorant E, van den Brandt PA, Goldbohm RA. A prospective cohort study on the relationship between onion and leek consumption, garlic supplement use and the risk of colorectal carcinoma in The Netherlands. Carcinogenesis. 1996;17:477–484. doi: 10.1093/carcin/17.3.477. [DOI] [PubMed] [Google Scholar]
- 43.Wang W, Dong Z, Zhang X, Li W, Li P, Chen X. Dietary and the risk of sporadic colorectal cancer in China: A case-control study. Iran J Public Health. 2018;47:1327–1335. [PMC free article] [PubMed] [Google Scholar]
- 44.Levi F, Pasche C, La Vecchia C, Lucchini F, Franceschi S. Food groups and colorectal cancer risk. Br J Cancer. 1999;79:1283–1287. doi: 10.1038/sj.bjc.6690206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Steinmetz KA, Kushi LH, Bostick RM, Folsom AR, Potter JD. Vegetables, fruit, and colon cancer in the Iowa women's health study. Am J Epidemiol. 1994;139:1–15. doi: 10.1093/oxfordjournals.aje.a116921. [DOI] [PubMed] [Google Scholar]
- 46.Franceschi S, Parpinel M, La Vecchia C, Favero A, Talamini R, Negri E. Role of different types of vegetables and fruit in the prevention of cancer of the colon, rectum, and breast. Epidemiology. 1998;9:338–341. doi: 10.1097/00001648-199805000-00020. [DOI] [PubMed] [Google Scholar]
- 47.Yuan P, Lin L, Zheng K, Wang W, Wu S, Huang L, Wu B, Chen T, Li X, Cai L. Risk factors for gastric cancer and related serological levels in Fujian, China: hospital-based case-control study. BMJ Open. 2020;10:e042341. doi: 10.1136/bmjopen-2020-042341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Galeone C, Pelucchi C, Levi F, Negri E, Franceschi S, Talamini R, Giacosa A, La Vecchia C. Onion and garlic use and human cancer. Am J Clin Nutr. 2006;84:1027–1032. doi: 10.1093/ajcn/84.5.1027. [DOI] [PubMed] [Google Scholar]
- 49.Witte JS, Longnecker MP, Bird CL, Lee ER, Frankl HD, Haile RW. Relation of vegetable, fruit, and grain consumption to colorectal adenomatous polyps. Am J Epidemiol. 1996;144:1015–1025. doi: 10.1093/oxfordjournals.aje.a008872. [DOI] [PubMed] [Google Scholar]
- 50.Annema N, Heyworth JS, McNaughton SA, Iacopetta B, Fritschi L. Fruit and vegetable consumption and the risk of proximal colon, distal colon, and rectal cancers in a case-control study in Western Australia. J Am Diet Assoc. 2011;111:1479–1490. doi: 10.1016/j.jada.2011.07.008. [DOI] [PubMed] [Google Scholar]
- 51.Myneni AA, Chang SC, Niu R, Liu L, Swanson MK, Li J, Su J, Giovino GA, Yu S, Zhang ZF, Mu L. Raw garlic consumption and lung cancer in a Chinese population. Cancer Epidemiol Biomarkers Prev. 2016;25:624–633. doi: 10.1158/1055-9965.EPI-15-0760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shi X, Lv Y, Mao C, Yuan J, Yin Z, Gao X, Zhang Z. Garlic consumption and all-cause mortality among Chinese oldest-old individuals: A population-based cohort study. Nutrients. 2019;11:1504. doi: 10.3390/nu11071504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Powell JT, Sweeting MJ. Retrospective studies. Eur J Vasc Endovasc Surg. 2015;50:675. doi: 10.1016/j.ejvs.2015.07.005. [DOI] [PubMed] [Google Scholar]
- 54.Yin X, Zhang R, Feng C, Zhang J, Liu D, Xu K, Wang X, Zhang S, Li Z, Liu X, Ma H. Diallyl disulfide induces G2/M arrest and promotes apoptosis through the p53/p21 and MEK-ERK pathways in human esophageal squamous cell carcinoma. Oncol Rep. 2014;32:1748–1756. doi: 10.3892/or.2014.3361. [DOI] [PubMed] [Google Scholar]
- 55.Choi YH. Diallyl trisulfide induces apoptosis and mitotic arrest in AGS human gastric carcinoma cells through reactive oxygen species-mediated activation of AMP-activated protein kinase. Biomed Pharmacother. 2017;94:63–71. doi: 10.1016/j.biopha.2017.07.055. [DOI] [PubMed] [Google Scholar]
- 56.Xiao D, Pinto JT, Gundersen GG, Weinstein IB. Effects of a series of organosulfur compounds on mitotic arrest and induction of apoptosis in colon cancer cells. Mol Cancer Ther. 2005;4:1388–1398. doi: 10.1158/1535-7163.MCT-05-0152. [DOI] [PubMed] [Google Scholar]
- 57.Shang A, Cao SY, Xu XY, Gan RY, Tang GY, Corke H, Mavumengwana V, Li HB. Bioactive compounds and biological functions of garlic (Allium sativum L.) Foods. 2019;8:246. doi: 10.3390/foods8070246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yin X, Zhang J, Li X, Liu D, Feng C, Liang R, Zhuang K, Cai C, Xue X, Jing F, et al. DADS suppresses human esophageal xenograft tumors through RAF/MEK/ERK and mitochondria-dependent pathways. Int J Mol Sci. 2014;15:12422–12441. doi: 10.3390/ijms150712422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang W, Ha M, Gong Y, Xu Y, Dong N, Yuan Y. Allicin induces apoptosis in gastric cancer cells through activation of both extrinsic and intrinsic pathways. Oncol Rep. 2010;24:1585–1592. doi: 10.3892/or_00001021. [DOI] [PubMed] [Google Scholar]
- 60.Zhang X, Zhu Y, Duan W, Feng C, He X. Allicin induces apoptosis of the MGC-803 human gastric carcinoma cell line through the p38 mitogen-activated protein kinase/caspase-3 signaling pathway. Mol Med Rep. 2015;11:2755–2760. doi: 10.3892/mmr.2014.3109. [DOI] [PubMed] [Google Scholar]
- 61.Park SY, Cho SJ, Kwon HC, Lee KR, Rhee DK, Pyo S. Caspase-independent cell death by allicin in human epithelial carcinoma cells: Involvement of PKA. Cancer Lett. 2005;224:123–132. doi: 10.1016/j.canlet.2004.10.009. [DOI] [PubMed] [Google Scholar]
- 62.Xiao D, Pinto JT, Soh JW, Deguchi A, Gundersen GG, Palazzo AF, Yoon JT, Shirin H, Weinstein IB. Induction of apoptosis by the garlic-derived compound S-allylmercaptocysteine (SAMC) is associated with microtubule depolymerization and c-Jun NH(2)-terminal kinase 1 activation. Cancer Res. 2003;63:6825–6837. [PubMed] [Google Scholar]
- 63.Jiang X, Zhu X, Huang W, Xu H, Zhao Z, Li S, Li S, Cai J, Cao J. Garlic-derived organosulfur compound exerts antitumor efficacy via activation of MAPK pathway and modulation of cytokines in SGC-7901 tumor-bearing mice. Int Immunopharmacol. 2017;48:135–145. doi: 10.1016/j.intimp.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 64.Zhu X, Jiang X, Li A, Sun Y, Liu Y, Sun X, Feng X, Li S, Zhao Z. S-allylmercaptocysteine suppresses the growth of human gastric cancer xenografts through induction of apoptosis and regulation of MAPK and PI3K/Akt signaling pathways. Biochem Biophys Res Commun. 2017;491:821–826. doi: 10.1016/j.bbrc.2017.06.107. [DOI] [PubMed] [Google Scholar]
- 65.Yan JY, Tian FM, Hu WN, Zhang JH, Cai HF, Li N. Apoptosis of human gastric cancer cells line SGC 7901 induced by garlic-derived compound S-allylmercaptocysteine (SAMC) Eur Rev Med Pharmacol Sci. 2013;17:745–751. [PubMed] [Google Scholar]
- 66.Li X, Ni J, Tang Y, Wang X, Tang H, Li H, Zhang S, Shen X. Allicin inhibits mouse colorectal tumorigenesis through suppressing the activation of STAT3 signaling pathway. Nat Prod Res. 2019;33:2722–2725. doi: 10.1080/14786419.2018.1465425. [DOI] [PubMed] [Google Scholar]
- 67.Bat-Chen W, Golan T, Peri I, Ludmer Z, Schwartz B. Allicin purified from fresh garlic cloves induces apoptosis in colon cancer cells via Nrf2. Nutr Cancer. 2010;62:947–957. doi: 10.1080/01635581.2010.509837. [DOI] [PubMed] [Google Scholar]
- 68.Yin X, Feng C, Han L, Ma Y, Jiao Y, Wang J, Jia L, Jing F, Gao X, Zhang Y, Zhang J. Diallyl disulfide inhibits the metastasis of type II esophageal-gastric junction adenocarcinoma cells via NF-κB and PI3K/AKT signaling pathways in vitro. Oncol Rep. 2018;39:784–794. doi: 10.3892/or.2017.6113. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Data
Supporting Data
Data Availability Statement
The study was registered in PROSPERO (CRD42020179464).