Mutant HER2 needs mutant HER3 to be an effective oncogene - PubMed (original) (raw)
Comment
Mutant HER2 needs mutant HER3 to be an effective oncogene
Raphael Trenker et al. Cell Rep Med. 2021.
Abstract
Hanker et al. reveal that co-occurring missense mutations in the human epidermal growth factor receptor 2 (HER2) and its catalytically inactive homolog HER3 synergize to promote oncogenic signaling by the HER2/HER3 complex.
© 2021 The Author(s).
Conflict of interest statement
N.J. is a member of the SAB and a shareholder of Turning Point Therapeutics, SUDO Biosciences, and Type6 Therapeutics. The Jura laboratory has received sponsored research support from Genentech. Other authors do not declare competing interests.
Figures
Figure 1
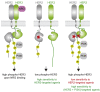
Co-occurring gain-of-function mutations in HER2 and HER3 promote their dimerization resulting in kinase activation and conferring resistance to HER2-targeting agents Left, upon neuregulin (NRG) growth factor stimulation, HER2 and HER3 assemble into an active heterocomplex that is required for HER3 phosphorylation and efficient activation of the PI3K/AKT pathway. Middle, oncogenic HER2 missense mutations discussed by Hanker and colleagues shift equilibrium toward the active conformation of the HER2 kinase but are ineffective in driving formation of the stable complexes of mutant HER2 with wild-type HER3 in the absence of NRG. In this scenario, sensitivity to HER2-targeted agents is maintained. Right, co-occurring mutations in HER2 and HER3 receptors drive NRG-independent receptor association (only kinase domain mutations are depicted here), resulting in robust HER3 phosphorylation and activation of the PI3K/Akt pathway. This leads to cellular transformation, invasiveness, and resistance to HER2-targeted therapeutics, such as neratinib. Combination of HER2-targeted agents with PI3K inhibitors is effective in blocking oncogenic signaling in the presence of the composite HER2 and HER3 mutations.
Comment on
- Co-occurring gain-of-function mutations in HER2 and HER3 modulate HER2/HER3 activation, oncogenesis, and HER2 inhibitor sensitivity.
Hanker AB, Brown BP, Meiler J, Marín A, Jayanthan HS, Ye D, Lin CC, Akamatsu H, Lee KM, Chatterjee S, Sudhan DR, Servetto A, Brewer MR, Koch JP, Sheehan JH, He J, Lalani AS, Arteaga CL. Hanker AB, et al. Cancer Cell. 2021 Aug 9;39(8):1099-1114.e8. doi: 10.1016/j.ccell.2021.06.001. Epub 2021 Jun 24. Cancer Cell. 2021. PMID: 34171264 Free PMC article.
References
- Bose R., Kavuri S.M., Searleman A.C., Shen W., Shen D., Koboldt D.C., Monsey J., Goel N., Aronson A.B., Li S. Activating HER2 mutations in HER2 gene amplification negative breast cancer. Cancer Discov. 2013;3:224–237. doi: 10.1158/2159-8290.CD-12-0349. -DOI -PMC -PubMed
- Hanker, Brewer, A.B., M.R., J.H., Sheehan, J.H., Koch, J.P., Sliwoski, G.R., Nagy, R., Lanman, R., Berger, M.F., Hyman, D.M., Solit, D.B., et al. (2017). An Acquired HER2(T798I) Gatekeeper Mutation Induces Resistance to Neratinib in a Patient with HER2 Mutant-Driven Breast Cancer. Cancer Discov. 2017, 575–585. 10.1158/2159-8290.CD-16-1431. -PMC -PubMed
- Smyth L.M., Piha-Paul S.A., Won H.H., Schram A.M., Saura C., Loi S., Lu J., Shapiro G.I., Juric D., Mayer I.A. Efficacy and Determinants of Response to HER Kinase Inhibition in HER2-Mutant Metastatic Breast Cancer. Cancer Discov. 2020;10:198–213. doi: 10.1158/2159-8290.CD-19-0966. -DOI -PMC -PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous