Chemoenzymatic Syntheses of Fluorine-18-Labeled Disaccharides from [18F] FDG Yield Potent Sensors of Living Bacteria In Vivo - PubMed (original) (raw)
. 2023 Aug 16;145(32):17632-17642.
doi: 10.1021/jacs.3c03338. Epub 2023 Aug 3.
Marina López-Álvarez 1, Sarah J Rabbitt 1, Aryn A Alanizi 1, Rebecca Shuere 1, Kondapa Naidu Bobba 1, Joseph Blecha 1, Sasank Sakhamuri 1, Michael J Evans 1, Kenneth W Bayles 2, Robert R Flavell 1, Oren S Rosenberg 3, Renuka Sriram 4, Tom Desmet 4, Bernd Nidetzky 5, Joanne Engel 4, Michael A Ohliger 1 6, James S Fraser 7, David M Wilson 1
Affiliations
- PMID: 37535945
- PMCID: PMC10436271
- DOI: 10.1021/jacs.3c03338
Chemoenzymatic Syntheses of Fluorine-18-Labeled Disaccharides from [18F] FDG Yield Potent Sensors of Living Bacteria In Vivo
Alexandre M Sorlin et al. J Am Chem Soc. 2023.
Abstract
Chemoenzymatic techniques have been applied extensively to pharmaceutical development, most effectively when routine synthetic methods fail. The regioselective and stereoselective construction of structurally complex glycans is an elegant application of this approach that is seldom applied to positron emission tomography (PET) tracers. We sought a method to dimerize 2-deoxy-[18F]-fluoro-d-glucose ([18F]FDG), the most common tracer used in clinical imaging, to form [18F]-labeled disaccharides for detecting microorganisms in vivo based on their bacteria-specific glycan incorporation. When [18F]FDG was reacted with β-d-glucose-1-phosphate in the presence of maltose phosphorylase, the α-1,4- and α-1,3-linked products 2-deoxy-[18F]-fluoro-maltose ([18F]FDM) and 2-deoxy-2-[18F]-fluoro-sakebiose ([18F]FSK) were obtained. This method was further extended with the use of trehalose (α,α-1,1), laminaribiose (β-1,3), and cellobiose (β-1,4) phosphorylases to synthesize 2-deoxy-2-[18F]fluoro-trehalose ([18F]FDT), 2-deoxy-2-[18F]fluoro-laminaribiose ([18F]FDL), and 2-deoxy-2-[18F]fluoro-cellobiose ([18F]FDC). We subsequently tested [18F]FDM and [18F]FSK in vitro, showing accumulation by several clinically relevant pathogens including Staphylococcus aureus and Acinetobacter baumannii, and demonstrated their specific uptake in vivo. Both [18F]FDM and [18F]FSK were stable in human serum with high accumulation in preclinical infection models. The synthetic ease and high sensitivity of [18F]FDM and [18F]FSK to S. aureus including methicillin-resistant (MRSA) strains strongly justify clinical translation of these tracers to infected patients. Furthermore, this work suggests that chemoenzymatic radiosyntheses of complex [18F]FDG-derived oligomers will afford a wide array of PET radiotracers for infectious and oncologic applications.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Figure 1
(A) Partial list of disaccharide phosphorylases with the potential for the chemoenzymatic synthesis of disaccharides via reverse phosphorolysis. (B) Reverse phosphorolysis of substrates using disaccharide phosphorylase with either α- or β-glucose-1-phosphate.
Figure 2
Radiochemical syntheses of 2-deoxy-2-[18F]fluoro-maltose ([18F]FDM) and 2-deoxy-2-[18F]fluoro-sakebiose ([18F]FSK). (A) Synthesis of the β-
d
-glucose-1-phosphate (βGlc1-P) precursor starting from acetobromo-α-
d
-glucose (S1). (B) Enzymatic radiosynthesis of [18F]FDM and [18F]FSK from 2-deoxy-2-[18F]fluoro-
d
-glucose [18F]FDG and βGlc1-P using maltose phosphorylase. (C) Radio HPLC analysis of crude products using a YMC-Pack Polyamine II column.
Figure 3
(A) Chemoenzymatic radiosyntheses of [18F]FDT, [18F]FDL, [18F]FDC, [18F]FDM, and [18F]FSK from [18F]FDG. All reactions were carried out at 37 °C, stirring for 20 min, using 6 mg (0.020 mmol) of precursor, 0.3 mg of enzyme (3–6 units), and 10–15 mCi [18F]FDG in 0.5 mL of citrate buffer (0.1 M, pH = 6.0). (B) Radio HPLC analysis of each enzymatic reaction using a YMC-Pack Polyamine II column.
Figure 4
(A) In vitro bacteria uptake for [18F]FDG, 2-deoxy-2-[18F]-fluoro-
d
-sorbitol ([18F]FDS), [18F]FDM, and [18F]FSK. (B) In vitro bacteria uptake of [18F]FDM and [18F]FSK in Gram-positive and Gram-negative pathogens. (C) Accumulation of [18F]FDM and [18F]FSK in S. aureus with increasing concentrations of unlabeled maltose and sakebiose, respectively. (D) In vitro bacteria uptake of [18F]FDM and [18F]FSK in methicillin-resistant S. aureus (MRSA) clinical strains.
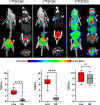
Figure 5
μPET-CT imaging of MRSA myositis in mice with [18F]FDM, [18F]FSK, and [18F]FDG. The red arrows indicate the site of inoculation with live bacteria, while the white arrows correspond to heat-killed bacteria. The corresponding bar graphs indicate region-of-interest (ROI) analysis. As reflected by the images, the mean [18F]FDM and [18F]FSK accumulation for tissues infected with live bacteria was respectively 6.1- and 6.5-fold higher than that seen for heat-killed inoculation (P < 0.0001). In contrast, this difference was not seen for [18F]FDG.
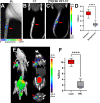
Figure 6
Imaging of S. aureus in vertebral discitis-osteomyelitis (VDO) rat models and A. baumannii in a myositis mouse model using [18F]FSK. (A) Optical tomography image of a rat tail showing the bioluminescent signal from S. aureus Xen29 inoculation. (B) Computed tomography study performed at 10 days highlights the similarity between rodent and human discitis osteomyelitis. (C) PET/CT imaging of S. aureus Xen29 vertebral discitis-osteomyelitis (VDO) in rat (N = 5) with [18F]FSK. (D) ROI analysis showing increased signal in segments inoculated with live bacteria versus background (P < 0.0001). (E) PET/CT imaging of A. baumannii myositis in mice (N = 6) with [18F]FSK. The red arrows indicate the site of inoculation with live bacteria, while the white arrows correspond to heat-killed bacteria. (F) ROI analysis showing an increased signal in infected muscle versus inflammation (P < 0.0001).
Update of
- Chemoenzymatic syntheses of fluorine-18-labeled disaccharides from [ 18 F]FDG yield potent sensors of living bacteria in vivo.
Sorlin AM, López-Álvarez M, Rabbitt SJ, Alanizi AA, Shuere R, Bobba KN, Blecha J, Sakhamuri S, Evans MJ, Bayles KW, Flavell RR, Rosenberg OS, Sriram R, Desmet T, Nidetzky B, Engel J, Ohliger MA, Fraser JS, Wilson DM. Sorlin AM, et al. bioRxiv [Preprint]. 2023 May 20:2023.05.20.541529. doi: 10.1101/2023.05.20.541529. bioRxiv. 2023. PMID: 37293043 Free PMC article. Updated. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 EB024014/EB/NIBIB NIH HHS/United States
- R01 EB025985/EB/NIBIB NIH HHS/United States
- R01 EB030897/EB/NIBIB NIH HHS/United States
- R21 AI164684/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources