Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major (original) (raw)
Main
Vaccines have made a substantial global impact on morbidity and mortality of a variety of bacterial and viral infections. Nevertheless, there are no licensed vaccines that are protective against HIV, malaria or pulmonary tuberculosis infection. For these pathogens, the cellular immune response comprising CD4+ T cells, CD8+ T cells or both can be important in controlling infection and preventing or delaying the onset of disease. Thus vaccine development for these infections is focused on generating protective T-cell responses1. A key mechanism by which T cells mediate their effector function is through the production of various cytokines. However, due to the heterogeneity of T-cell cytokine responses generated by different vaccines, there are still no well defined immune correlates of protection for infections requiring T-cell responses. Therefore a crucial step in vaccine development requires improved understanding of the functional heterogeneity of T-cell cytokine responses.
After activation, naive CD4+ T cells can differentiate into functional subsets termed TH1 or TH2 cells2. TH1 responses are required to mediate protection against a variety of intracellular infections. Such responses consist of populations of cells that secrete interferon (IFN)-γ, tumor necrosis factor (TNF) or interleukin (IL)-2 in various combinations3,4,5,6. Differences in the types of cytokines produced by individual cells have profound implications for their capacity to mediate effector function, be sustained as memory cells or both. In this regard, CD4+ T cells that secrete only IFN-γ have limited capacity to develop into memory cells compared with IL-2- or IL-2- and IFN-γ-producing cells6,7,8. This implies that, for example, vaccines eliciting a high frequency of single-positive IFN-γ producing cells may be limited in their ability to provide durable protection. The premise that distinct populations of TH1 responses determine vaccine efficacy provides a conceptual framework to define an immune correlate of protection.
Most vaccine studies for infections requiring TH1 responses measure the frequency of IFN-γ producing cells as the primary immune correlate of protection9. Although IFN-γ is clearly necessary10,11,12, using it as a single immune parameter may not always be sufficient to predict protection13,14,15,16. TNF is another effector cytokine that can mediate control of intracellular infections17,18. Indeed, IFN-γ and TNF synergize in their capacity to mediate killing of pathogens19,20. IL-2 has little direct effector function but strongly enhances the expansion of CD4+ and CD8+ T cells, leading to a more efficient effector response. Thus, we hypothesized that CD4+ T cells that are multifunctional and simultaneously produce IFN-γ, TNF and IL-2 may provide optimal effector function and protection.
The best technique for assessing multiple functions of T cells simultaneously is multiparameter flow cytometry21. By assessing all combinations of IFN-γ, TNF and IL-2 at the single-cell level, one can define the quality of the CD4+ T-cell cytokine response. In the present study we used an experimental infection model in which different vaccines elicited qualitatively distinct TH1 responses and conferred varying degrees of protection. Our results showed that the frequency of multifunctional TH1 cells simultaneously secreting IFN-γ, TNF and IL-2 correlated best with protection. Notably, such cells seem specialized to secrete high amounts of IFN-γ and TNF. These data provide fundamental insight into how distinct populations of cytokine-producing cells are optimized for effector function and demonstrate a correlate of protection for vaccines targeting T-cell responses based on the quality of the response.
Results
Vaccine-elicited protection against Leishmania major
Models of L. major infection have been extensively used to understand the generation and maintenance of TH1 responses in the context of both natural infection and vaccine-elicited protection22. Infection of C57BL/6 mice mimics human disease, with the development of cutaneous lesions that are resolved by the host TH1 response through effector cytokines such as IFN-γ and TNF. To study the functional heterogeneity of primary TH1 responses generated in vivo and to define immune correlates of protection, we used a variety of vaccine formulations in the L. major model. We compared live vaccination, the gold standard for life-long protection in mice23,24 and humans25; or a recombinant leishmanial protein (MML) and a toll-like receptor (TLR) ligand (CpG), another formulation that confers protection in mice26,27; to replication-defective adenovirus that expresses MML (MML-ADV). Although not yet tested in the L. major model, adenoviral vaccines28,29 elicit strong T-cell responses and have potentially broad application for use in HIV30, malaria31 and tuberculosis32.
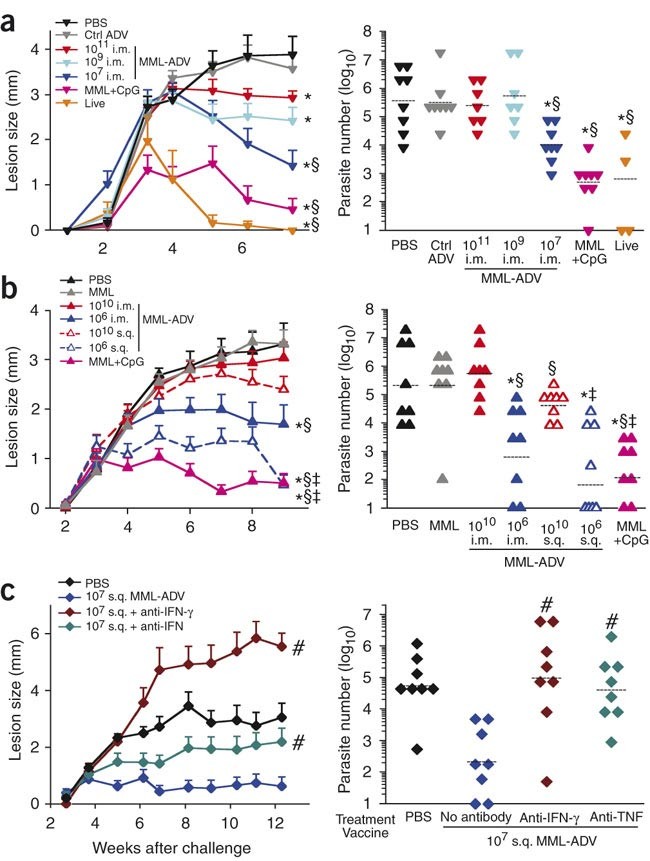
As a measure of protection, we compared lesion development and parasite burden after intradermal ear challenge with live L. major 1 month after immunization. The greatest degree of protection was observed in mice given the live and MML+CpG vaccines (Fig. 1a). Notably, we observed an inverse correlation between the dose of intramuscular (i.m.) MML-ADV and the level of protection; although all doses of MML-ADV resulted in smaller lesions compared with those in unvaccinated mice, low-dose MML-ADV (107) protected mice to the greatest degree (Fig. 1a). In a separate experiment, we observed a similar inverse correlation when MML-ADV was administered subcutaneously (s.q.; Fig. 1b), and the s.q. route consistently elicited better protection than the i.m. route at the same dose. The enhanced protection by s.q. immunization may reflect induction of skin-homing T cells or differences in the frequency or type of antigen-presenting cells by this route. A key finding is that less protection was observed with high-dose MML-ADV immunization, irrespective of route.
Figure 1: Vaccine-elicited protection against L. major.
Mice were challenged in the ear with live L. major after immunization with a variety of vaccine formulations. Left panels, ear lesion diameter over time (mean ± s.e.m., n ≥ 12). Right panels, number of parasites per ear in each vaccine group (lines are geometric means). (a) MML-ADV (i.m.) dose dependence compared with negative (PBS, control adenovirus (Ctrl ADV)) and positive (MML+CpG, live parasite) controls. (b) Comparison of i.m. versus s.q. at high (1010) or low (106) MML-ADV dose. (c) Requirement of IFN-γ or TNF for mediation of vaccine-elicited protection. Mice, vaccinated with 107 MML-ADV, received antibodies to IFN-γ or TNF at the time of challenge. Shown are representative data from three of nine experiments. Symbols indicate statistical differences for lesion sizes or endpoint parasite numbers, comparing with PBS (*P < 0.04), with 1011 or 1010 i.m. (§P < 0.03), with 1010 s.q. (‡P ≤ 0.02) or with 107 s.q. (#P ≤ 0.002).
The requirement for TH1 cells in mediating protection in vivo was assessed in several ways. First, vaccine-elicited protection was completely abrogated upon depletion of CD4+ T cells at the time of infection (data not shown). Next, depletion of IFN-γ or, to a lesser extent, TNF at the time of infection (Fig. 1c) abolished vaccine-mediated protection. Hence, CD4+ T cells, IFN-γ and TNF were required for vaccine-mediated protection in this model.
Functional heterogeneity among TH1 responses
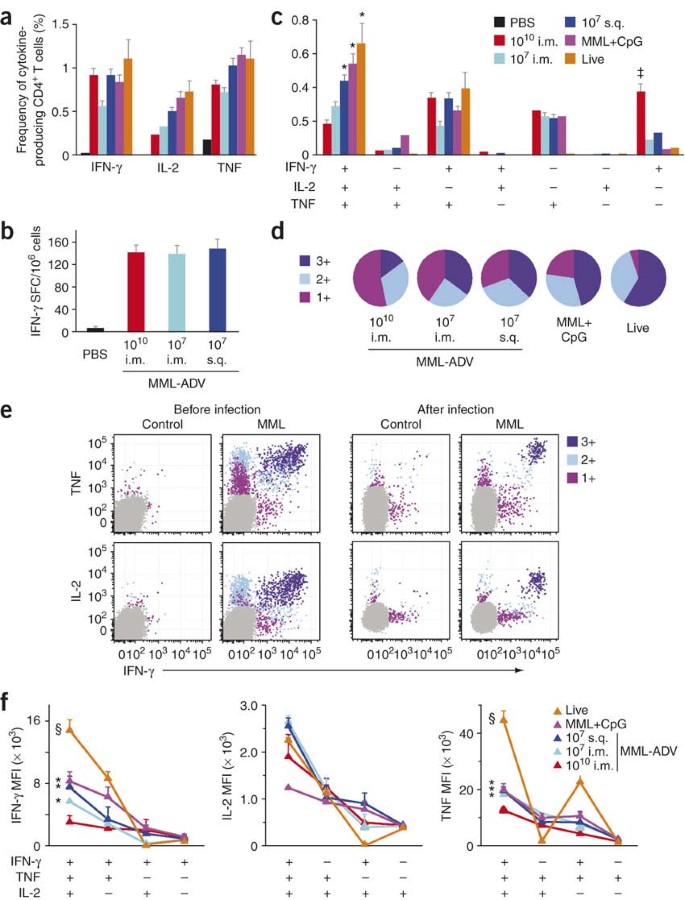
Based on the requirement for IFN-γ and role for TNF as effector cytokines that mediate protection, we assessed the frequency of antigen-specific IFN-γ-, IL-2- and TNF-producing CD4+ T cells at time of infection (Fig. 2a). One month after vaccination there was no difference between vaccine groups in the frequency of IFN-γ-producing cells in the spleen as assessed by intracellular cytokine staining (Fig. 2a) or enzyme-linked immunosorbent spot (ELISPOT) analysis (Fig. 2b). Note that the frequency of antigen-specific responses in live-vaccinated mice is under-represented, as all vaccine groups were restimulated with MML protein; using a complete spectrum of leishmanial antigens, cytokine responses after live vaccination are ∼15–20% (data not shown). Collectively, these data show that the total frequency of antigen-specific IFN-γ+ cells was not predictive of vaccine-elicited protection. Furthermore, protective efficacy was not related to differences in the breadth of the CD4+ T-cell response among the different vaccine formulations or to enhanced production of IL-4 or IL-13 in mice immunized with high-dose MML-ADV. We also looked for a potential role for IL-10 and regulatory T cells, but these did not seem to demonstrably affect the protection after high-dose MML-ADV vaccination (Supplementary Fig. 1 online).
Figure 2: Multifunctional TH1 cells are optimal effector cells.
(a–f) MML-specific cytokine production from splenocytes of vaccinated mice determined at the time of challenge. Multiparameter flow cytometry (Supplementary Fig. 2) was used to determine (a) the total frequency of IFN-γ-, IL-2- or TNF-producing CD4+ T cells, (c) the frequency of cells expressing each of the seven possible combinations of IFN-γ, IL-2 and TNF and (d) the fraction of the total response comprising cells expressing all three cytokines (3+), any two cytokines (2+), or any one cytokine (1+). (b) The number of IFN-γ spot forming cells (SFC) as measured by ELISPOT assay. (e) Representative fluorescence intensity distributions of 3+, 2+, and 1+ cells in a vaccinated mouse before and after infection (arbitrary units). (f) The IFN-γ, IL-2, and TNF MFIs of antigen-specific 3+, 2+, and 1+ cells for each vaccine group. Shown is the mean ± s.e.m. (n = 3 or 4) for one of at least three experiments. *Different from 1010 i.m. (*P ≤ 0.04); §,‡different from all groups (‡P ≤ 0.05, §P ≤ 0.004).
Based on the importance of IFN-γ and TNF in mediating protection (Fig. 1c), we undertook a more comprehensive functional analysis of cytokine-producing CD4+ T cells. Using multiparameter flow cytometry, seven distinct populations of cytokine-producing cells can be delineated at the single-cell level based on any combination of IFN-γ, IL-2 or TNF (Supplementary Fig. 2 online). The relative frequency of these distinct populations defines the quality of the TH1 response. Thus for IFN-γ, the total frequency of IFN-γ-producing cells encompasses four distinct populations: IFN-γ single-positive, IFN-γ+IL-2+, IFN-γ+TNF+ and IFNγ+IL-2+TNF+. In contrast to the lack of correlation seen by measuring the total frequency of IFN-γ producing cells as a single parameter (Fig. 2a,b), this analysis showed a correlation between the frequency of multifunctional (IFN-γ, IL-2 and TNF triple-positive) CD4+ T cells and the degree of protection in mice vaccinated with low-dose MML-ADV, MML+CpG or live L. major (Fig. 2c). Conversely, mice vaccinated with 1010 MML-ADV were not protected from challenge (Fig. 1b) despite having a high frequency of IFN-γ single-positive cells (Fig. 2c). Differences in the quality of the response between vaccine groups are represented pictorially by pie charts (Fig. 2d). Quantifying the fraction of the total cytokine response comprising three (3+), any two (2+) or any one (1+) cytokine, we found that over half of the response in the 1010 MML-ADV group was 1+ cells, whereas the response after live vaccination was dominated by multifunctional (3+) cells. The quality of the response in the draining lymph node and lung showed similar differences among the vaccine groups (Supplementary Fig. 3 online). Therefore, although different vaccines elicited similar frequencies of total antigen-specific IFN-γ responses, protection improved as the responses shifted qualitatively from 1+ to 3+.
There are several mechanisms by which 3+ cells could mediate enhanced protection. First, simultaneous production of both IFN-γ and TNF could lead to more efficient killing of parasites, and IL-2 could enhance the expansion of such cells. However, we did not observe proliferative differences between total MML-specific CD4+ T cells from mice vaccinated with high- or low-dose MML-ADV in vitro (Supplementary Fig. 4 online). A second possibility is that these cells may produce more of each cytokine on a per-cell basis. In this regard, there is substantial heterogeneity in the median fluorescence intensity (MFI) of the various cytokines, reflecting differences in cytokine production among subsets. Indeed, the MFI of 3+ cells was much higher for IFN-γ, IL-2 and TNF than all 2+ or 1+ cells after vaccination as well as during an ongoing infection (Fig. 2e). Notably, we observed a progressive decrease in the MFI for all cytokines as the degree of functionality decreased (3+ to 1+), irrespective of vaccine group, in spleen (Fig. 2f) as well as lymph node and lung (Supplementary Fig. 5 online). Overall, these data highlight a fundamental property of TH1 cytokine-producing cells by showing that functional differences existed at distinct stages of TH1 differentiation. They further imply that the increased secretion of cytokine by individual 3+ cells contributed to making them better effectors.
In comparing different vaccine formulations, we noted that the MFIs of 3+ cells elicited by different vaccines were not identical; indeed there was a hierarchy of cytokine production that correlates with protection (Fig. 2f). Multifunctional (3+) cells from mice vaccinated with live L. major had the highest MFI for IFN-γ and TNF compared with all other groups, whereas those from mice vaccinated with MML+CpG or 107 MML-ADV had a greater MFI for IFN-γ and TNF than those from the 1010 MML-ADV group. The enhanced MFI for IFN-γ and TNF from multifunctional cells from live-vaccinated mice may reflect the fact that cells from such mice are exposed to a low level of persistent antigen in vivo and are primed for optimal effector function. Hence, these data show differences in the potency of the effector cytokine responses even among discrete populations of TH1 cells.
Notably, in contrast to the MFIs for IFN-γ and TNF, there were no differences in the MFI for IL-2 in the multifunctional cells when comparing all the vaccine groups (Fig. 2f). Thus, different vaccines have a far greater influence on the relative potency of IFN-γ and TNF than IL-2 produced by multifunctional cells. Taken together, the quality of a response thus represented a multiplicative effect: responses from protected vaccine groups were more potent because they had a higher frequency of 3+ cells, each of which secreted more cytokine compared with cells from lesser- or unprotected vaccine groups.
Division of TH1 responses into Tcm and Tem subsets
Classification of T cells into central (Tcm) and effector memory (Tem) cells has been widely used to provide insight into the underlying functional capacity of such cells. Thus we characterized subsets of cytokine-producing cells as Tcm or Tem cells based on differences in CCR7 expression33 (Fig. 3). The majority of IFN-γ-producing cells (IFN-γ+IL-2+TNF+, IFN-γ+IL-2+, IFN-γ+TNF+ or IFN-γ+) in the spleen showed low CCR7 expression, whereas cells producing IL-2, TNF or both in the absence of IFN-γ had higher expression of CCR7. A similar distribution of cytokine-positive cells with respect to CCR7 expression was seen in lungs after vaccination with MML+CpG. Notably, in lymph nodes draining the site of immunization, there was a higher frequency of IL-2-, TNF-, or IL-2- and TNF-producing cells that had low expression of CCR7 compared with the frequency of such cells in spleen or lungs (Fig. 3). Finally, similar data were obtained from healed mice assessed 8 months after infection (data not shown). Thus, in this vaccine model, protection was best correlated with the induction of TH1 cells that were lower in expression of CCR7 and would be considered effector memory cells. Notably, such cells secrete IL-2 as well as TNF and IFN-γ.
Figure 3: CCR7 expression on distinct functional TH1 cells after vaccination.
Shown is the expression of CCR7 on total CD4+ T cells and on distinct functional antigen-specific TH1 cell populations (gated as in Supplementary Fig. 2) within the spleen (individual), lymph node (pooled) and lung (pooled) from various vaccine groups. Numbers represent the percentage of CCR7− cells within the CD4+ T-cell or functional cytokine subset. Plots represent spleens except where noted; data are representative of three independent experiments. γ, IFN-γ; 2, IL-2; T, TNF.
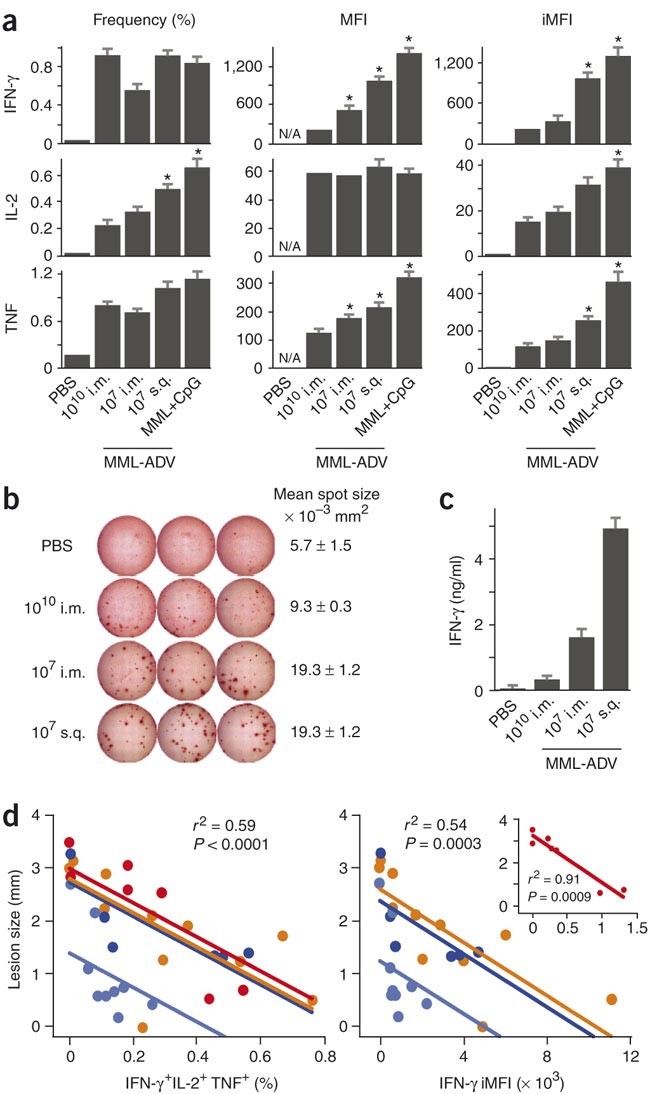
An immune correlate encompassing magnitude and quality
Differences in the frequency and MFI of 3+ cells between vaccine groups prompted us to develop a metric that incorporates both the magnitude and quality of a response and can be used to predict protection when comparing several vaccine formulations at the same time. The magnitude of a response is defined by the total frequency of CD4+ T cells producing a particular cytokine, whereas the MFI is used to assess the potency or quality of the response. By multiplying the frequency by the MFI, we derived a metric termed the integrated MFI (iMFI) that reflects the total functional response of a population of cytokine producing cells. Although there was little difference in the frequency of total IFN-γ-producing cells between vaccine groups, significant differences in the IFN-γ MFI resulted in iMFI values that were predictive of protective efficacy (Fig. 4a). The higher iMFI elicited by protective vaccines is determined by the underlying quality of the response and is likely to be driven by a high frequency of multifunctional cells (Fig. 2c) that produce more cytokine (Fig. 2f). As the iMFI reflects the total functional response, there should be an increase in the total amount of cytokine secreted during a response. Indeed, although the frequency of IFN-γ ELISPOTs between vaccine groups was the same (Fig. 2b), there was marked contrast in the size and intensity of the individual spot-forming cells (Fig. 4b), indicating that more IFN-γ per cell was produced in vaccine groups that elicited a greater degree of protection. Moreover, after in vitro stimulation, enhanced production of IFN-γ protein (Fig. 4c) correlated with the IFN-γ iMFI (Fig. 4a).
Figure 4: Prechallenge TH1 functionality predicts protection.
(a) For each cytokine, the total frequency (left), MFI (middle) and iMFI (right) of all IFN-γ-, IL-2- or TNF-producing antigen-specific CD4+ splenocytes 28 d after vaccination (mean ± s.e.m., n = 4 mice per group; *different from 1010 i.m., P ≤ 0.04). (b) Representative images of IFN-γ ELISPOT wells and mean spot size of IFN-γ ELISPOTs from the various vaccine groups. (c) MML-specific IFN-γ secretion measured by ELISA from splenocytes of vaccinated mice (mean ± s.e.m., n = 3). (d) The mean frequency of MML-specific multifunctional (3+) cells (left) and the IFN-γ iMFI (right) after vaccination both inversely correlate with the lesion size after challenge in four different experiments (represented by different colors). Each individual point represents a vaccine group that differs with respect to experiment, formulation, route, dose and time of challenge. Inset: data from an experiment done using an analog rather than digital instrument, accounting for the change in fluorescence scale. Aggregate statistical analysis across the three or four experiments yields a single correlation coefficient and significance value, computed from a multivariate linear least-squares model. In this model, the slope did not vary significantly by experiment, showing that, for example, for two groups of mice where the average 3+ population differs by 0.2%, we expect a difference in lesion size of 0.75 mm.
As the iMFI is a product of frequency and quality, protective iMFI values could also arise from a high frequency of lesser quality cells. Indeed, mice immunized with high- or low-dose MML-ADV had comparable IFN-γ iMFI at 10 d after immunization and showed similar protection when challenged at this time, despite differences in IFN-γ frequency (Supplementary Fig. 6 online). Finally, TNF and IL-2 iMFIs also correlated with protection (Fig. 4a). For TNF, this was due to the difference in MFI, as was seen with IFN-γ. By contrast, for IL-2, MFIs were similar in all vaccine groups, but the frequency varied. These data underscore the utility of iMFI for determining a total functional response and show that the iMFI for any of the cytokines can be useful for correlating with protection.
To determine a statistical correlation between immune protection and the magnitude and quality of the response, we plotted the mean frequency of 3+ TH1 cells at the time of challenge against the mean lesion size for four independent experiments incorporating a variety of vaccine formulations (MML-ADV, MML+CpG, live), time of challenge (10 d, 28 d), routes (i.m., s.q.) and MML-ADV doses (106–1011) (Fig. 4d, left). Across all variables, there was a strong inverse correlation between the frequency of 3+ cells and pathogenesis. Furthermore, in an example of how the iMFI can be a useful metric for individual cytokines, the IFN-γ iMFI significantly correlated with protection (Fig. 4d, right). Moreover, a similar relationship obtained using iMFI for TNF and to a lesser extent for IL-2 (data not shown). These findings were consistent with the fact that all three cytokines were in fact produced in highest amounts from the same multifunctional cells, cells which correlated with protection. In conclusion, these data provide two prospective immune correlates of protection that incorporate the quality of the response. The iMFI encompasses the total functional response of a given cytokine and, when assessed at the time of infection, provides a parameter for comparing the abilities of various vaccines to elicit protection. However, a full characterization of the quality of the response provides information on both function and phenotype, predicts protection and may offer insight into the durability of the response.
Finally, to assess how TH1 responses might mediate protection after vaccination, we performed an immune analysis at the site of infection after challenge (Supplementary Fig. 7 online). The results demonstrated maintenance of quality after challenge and highlighted the fact that live vaccination elicited a high frequency of multifunctional cells that migrate into the site of infection quickly.
Multifunctional TH1 responses elicited by BCG
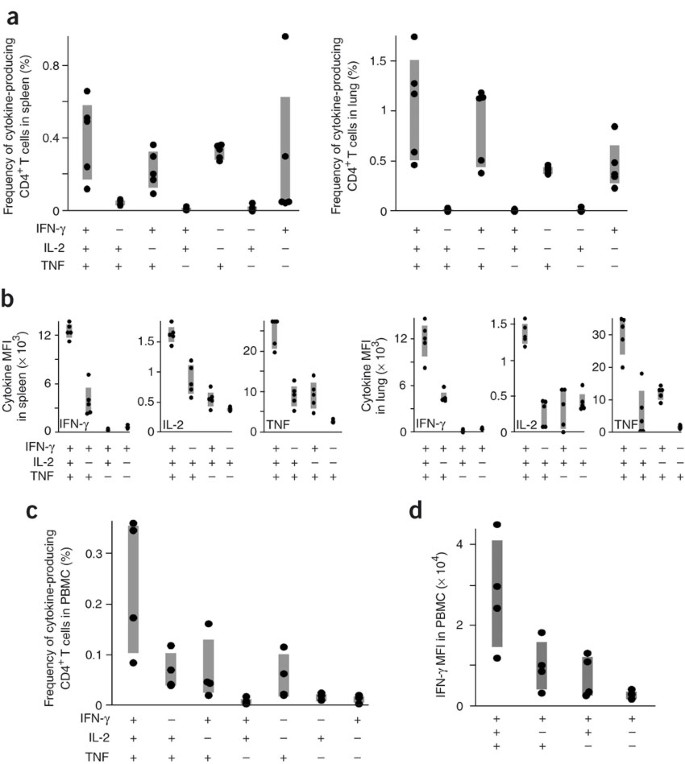
To determine whether the functional hierarchy of cytokine-producing TH1 cells existed in another vaccine model of infection requiring a TH1 response34, we assessed the quality of the TH1 response after Mycobacterium bovis bacillus Calmette-Guérin (BCG) immunization in mice and humans (Fig. 5). In mice, the antigen-specific TH1 response after BCG consisted of a high frequency of multifunctional (3+) and IFN-γ+TNF+ (2+) CD4+ T cells in both the spleen and the lung (Fig. 5a). Consistent with findings in the L. major model (Fig. 2f), there was a progressive decrease in the MFIs of IFN-γ, IL-2 and TNF when comparing 3+ cells with 2+ or 1+ cells (Fig. 5b). Upon aerosol challenge with M. tuberculosis, BCG vaccination conferred protection compared with the susceptibility of naive mice, as demonstrated by significantly decreased (P < 0.05) bacterial burdens (log10 colony forming units) at 1 month after challenge in both the spleen (naive, 5.22 ± 0.11; BCG, 4.41 ± 0.20) and lung (naive, 6.14 ± 0.07; BCG, 5.20 ± 0.06). In concert with the data from the mouse model, the major population of antigen-specific TH1 cells from peripheral blood mononuclear cells (PBMCs) of humans that received BCG immunization were multifunctional or dual IFN-γ- and TNF-producing cells (Fig. 5c). Moreover, the cytokine MFI hierarchy in humans was similar to that observed in mice when comparing multifunctional with single-positive cytokine-producing cells (Fig. 5d). Although these data are indicative that multifunctional TH1 cells may be of benefit in M. tuberculosis infection, a formal demonstration using different vaccine formulations encoding M. tuberculosis antigens that elicit distinct functional TH1 responses that confer varying protection will be required.
Figure 5: BCG vaccination elicits multifunctional TH1 cells in mice and humans.
(a) In mice (n = 4), the frequency of PPD-specific CD4+ T cells producing each possible cytokine combination in the spleen (left) or lung (right) 4 months after BCG vaccination. (b) MFIs of IFN-γ, IL-2, or TNF for 3+, 2+ and 1+ cells in mice after vaccination with BCG. (c) Multifunctional cytokine analysis and (d) IFN-γ MFI of PPD-specific, memory CD4+ T cells in PBMCs from four humans vaccinated with BCG at various times. Shaded bars, interquartile range. PPD responses in unvaccinated mice and humans were undetectable.
Discussion
This report reveals a fundamental property of TH1 cells, showing that there are distinct differences in the potency of effector cells demarcated by whether they secrete multiple cytokines. We show that TH1 cells that produce IL-2, TNF or both are mostly CCR7+ central memory cells. These populations can develop into IFN-γ-producing cells upon subsequent activation6 and are likely to provide a reservoir of long-term central memory cells. Although such populations have a role in protection35, they are unlikely to be sufficient for optimal protection in the absence of effector cells. The majority of CD4+ T cells that produce IL-2, IFN-γ and TNF, or IFN-γ and TNF were lower in expression of CCR7 and would be classified as effector memory cells. Based on their enhanced production of IFN-γ they are essential for mediating protection against L. major. Finally, single-positive CD4+ IFN-γ-producing cells have low MFIs and would be far less efficient as effector cells. Thus vaccines such as high-dose MML-ADV, in which single-positive IFN-γ producing cells constitute a substantial portion of the total CD4+ cytokine response, would be more limited in mediating optimal and sustained protection. These data raise the question of whether TH1 cells transition from multifunctional to single-positive IFN-γ-producing cells. Indeed, this type of linear progression as it relates to cytokine production has been suggested at the population level for CD8+ T cells in response to chronic viral infection36.
A formal demonstration that multifunctional cells directly mediate protection is not possible at this point because of the technical limitations in isolating enough viable cells that simultaneously produce three cytokines. Nevertheless, it has been shown that TH1 effector cells, as defined by CD62Llo expression, isolated from mice previously infected with L. major can confer protection upon transfer to naive mice35. Our findings that these effector cells are predominantly multifunctional provide evidence for their key role in protection. In conclusion, vaccines must generate sufficient, but not excessive, differentiation to ensure a population of TH1 responses that comprise protective effectors and durable memory cells.
The factors controlling the magnitude and quality of TH1 responses are the amount and duration of antigen37,38,39,40, the type of antigen-presenting cell41 and the innate cytokine milieu42 generated by the vaccine or adjuvant. A previous study in a mouse model of a persistent viral infection showed that CD4+ T cells undergo progressive exhaustion and ultimately become unresponsive43. As shown here, high-dose MML-ADV, which is likely to provide more antigen and innate cytokine production, elicited a substantially higher frequency of total IFN-γ-producing cells compared with low-dose MML-ADV at the peak of the response (10 d; Supplementary Fig. 6). However, by 28 d the frequency of the IFN-γ response had contracted to become comparable with that seen in other vaccine groups, but its quality was substantially different and yielded poor protection. These data indicate that the magnitude of the peak effector TH1 response may not necessarily correlate with durability and protection and that for TH1 cells 'more' is not always 'better'.
In terms of the durability of responses after immunization with MML-ADV or MML+CpG, we assessed the magnitude and quality over an extended period of time (Supplementary Fig. 8 online). Notably, the quality of the TH1 responses generated by MML-ADV or MML+CpG was established as early as 10–28 d after immunization and remained consistent over a prolonged period of time. Thus, vaccines seemed to imprint the quality of the response early after immunization. By contrast, the magnitude of the responses decreased over time in all vaccine groups (Supplementary Fig. 8). Although we have not determined whether low-dose MML-ADV provides long-term protection, the progressive decrease in the TH1 response makes it unlikely that this vaccine modality would elicit sustained protection. Moreover, we have observed that protection after MML+CpG begins to wane 6 months after immunization. Thus, despite a favorable quality, the magnitude of this response may not be sufficient to mediate optimal protection. A final point is that although the quality of responses may remain constant over a defined period of time, additional immunization(s) can alter the quality of the responses44.
In contrast to MML-ADV and MML+CpG vaccines, which do not provide persistent antigen and are unlikely to mediate sustained protection, vaccination or infection with live L. major elicits complete life-long immunity. As L. major has evolved mechanisms to allow persistence in host cells45, it provides a low level of sustained antigen, which is required for generating and sustaining a high frequency of multifunctional effector cells. This requirement for persistent antigen23,24 puts a substantial constraint on whether nonlive vaccines can generate a sufficient frequency of multifunctional TH1 effector cells that are sustained over a prolonged period of time. Thus in using nonlive recombinant vaccine formulations or live vaccines that are cleared from the body, boosting throughout the lifetime of the individual may be necessary to ensure full protective immunity for infections requiring TH1 responses. Boosting may help maintain a sufficient frequency of potent effector cells that act immediately upon infection as well as a reservoir of memory cells with effector capacity35.
In conclusion, this study provides evidence why multifunctional TH1 cells are better for mediating effector function. Although such cells may be necessary for optimal and sustained protection for infections requiring TH1 responses, they may not always be sufficient. This will depend on the magnitude, potency and durability of multifunctional responses and may vary depending on the vaccine. The findings that multifunctional TH1 cells are better effectors also extend to CD8+ T cells. Indeed, it has recently been shown that CD8+ T cells from HIV-infected individuals with better control of infection have an increased frequency of multifunctional cells compared with those individuals with progressive disease46. In this regard, we have also observed that CD8+ T cells that secrete IL-2, TNF and IFN-γ make more IFN-γ than IFN-γ+TNF+ or IFN-γ+ cells (data not shown), indicating that these multifunctional CD8+ T cells might be better effectors. Hence, in evaluating vaccine candidates against HIV, malaria and tuberculosis in which TH1 T cells, CD8+ T cells or both may be required, it will be important to determine which vaccine or regimen elicits the highest frequency of the most potent multifunctional cells. The findings reported here should be useful for improving the design of preventive and therapeutic vaccines against infections and cancer to elicit qualitatively better T-cell responses.
Methods
Mice.
C57BL/6 mice purchased from The Jackson Laboratory were maintained in the Vaccine Research Center Animal Care Unit under pathogen-free conditions. All experiments were approved by the Vaccine Research Center animal care and use committee.
Immunization.
MML, also known as Leish-111f, is a recombinant leishmanial polyprotein shown to be protective in vivo47. We immunized mice with 106–1011 viral particles (approximately 16 particles per infectious unit) of replication-deficient adenovirus that expressed MML (MML-ADV), in a single injection either i.m. (leg) or s.q. (foot). MML (25 μg) and CpG 1826 (Coley Pharmaceutical Group; 50 μg) were administered s.q. three times, 2 weeks apart, as previously described26. In some experiments, 1 mg of antibody to CD4 (GK1.5; Harlan Bioproducts for Science) was given intraperitoneally 2 d before and after vaccination. For live vaccinations, mice received 104 metacyclic L. major promastigotes (V1, MHOM/IL/80/Friedlin) s.q. or 5 × 105 M. bovis BCG Pasteur (Staten Serum Institute) i.m.
Infectious challenge and parasite quantification.
Mice were challenged intradermally in both ears with 500–1,000 metacyclic L. major promastigotes either 10 or 28 d after vaccination, as previously described48. Live-vaccinated mice had healed the infection 6 months earlier. In some experiments, we administered 0.5 mg antibody to IFN-γ (XMG1.2; Harlan Bioproducts for Science) or antibody to TNF (MP6-XT22) intraperitoneally at the time of infection and weekly thereafter. We measured the diameter of dermal lesions (at least 12 ears per time point) weekly. Parasite numbers in the ear were determined as previously described48 and scored as the highest dilution containing viable parasites after incubation for 5 d at 26 °C. For M. tuberculosis studies, naive or BCG-vaccinated mice were challenged with 200 virulent M. tuberculosis Erdman 3 months after immunization. We counted the number of colony-forming units to determine the bacterial burden in the spleen and lung 1 month after challenge.
ELISPOT and ELISA.
For individual spleen samples (n = 3), we incubated 2 × 105 (ELISPOT) or 4 × 105 (ELISA) cells in triplicate with 20 μg/ml MML protein in a total volume of 200 μl at 37 °C. The frequency of IFN-γ producing cells was measured after 24 h (BD ELISPOT; BD Pharmingen) using an Axioplan 2 imaging system (Zeiss) and IFN-γ protein in cell supernatants was measured after 48 h (Quantikine ELISA; R&D Systems) using a Spectramax Plus (Molecular Devices).
Multiparameter flow cytometry.
We incubated cells harvested from infected ears47 or 1.5 × 106 leukocytes from spleen, lymph node or lung with 2 μg/ml antibody to CD28 (BD Pharmingen) and 20 μg/ml MML or PPD (purified protein derivative, CSL Ltd.) for 2 h at 37 °C. Brefeldin A (BFA; Sigma-Aldrich) was added at a final concentration of 10 μg/ml and cells were incubated for an additional 4 h before intracellular cytokine staining. Cells were incubated with the viability dye ViViD (Molecular Probes) as previously described49, followed by staining for CD3, CD4, CD8 (Biolegend), CCR7 (Biolegend), IFN-γ, IL-2, TNF and IL-10 (eBioscience) using the BD Cytofix/Cytoperm kit according to the manufacturer's instructions. Finally, we resuspended cells in BD stabilizing fixative. All antibodies and all reagents for intracellular cytokine staining were purchased from BD Pharmingen except where noted. We acquired 250,000 live lymphocytes per sample using a modified BD LSR II flow cytometer and analyzed the data using SPICE 4.0 and FlowJo software (Tree Star). For additional information, see Supplementary Figure 2. Samples from one experiment were analyzed using a BD FACS Calibur; the change in instrument type (analog Calibur versus digital LSR) accounts for differences in the fluorescence scales and iMFI values between experiments (for example, Fig. 4d, right).
Human donors.
Informed consent was obtained from all subjects and the study was approved by the local Ethics Committee for Copenhagen and Frederiksberg (KF11-004/01) in 2001. PBMCs were re-stimulated with 5 μg/ml PPD in the presence of 1 μg/ml antibody to CD49d and antibody to CD28 in the presence of 10 μg/ml BFA for 6 h before staining for CD3, CD4, CD8, CD27, CD45RO, IFN-γ, IL-2 and TNF. Naive CD4+ T cells were identified as CD27+CD45RO−.
Statistics.
All comparisons between vaccine groups used a two-tailed Student's _t_-test assuming unequal variances. For challenge data, we compared lesions (n ≥ 12 ears per group) at the time of peak lesion size in unimmunized mice (∼7 weeks) and quantified parasite numbers at the end of the lesion time course. Comparison of frequency, MFI and iMFI in the spleen was based on 3–4 mice per group. All cytokine frequencies and iMFI values reported are after background subtraction of the frequency or iMFI of the identically gated population of cells from the same sample stimulated without antigen. We calculated correlations between the prechallenge mean frequency of 3+ cells or mean IFN-γ iMFI and the postchallenge mean lesion size (∼7 weeks) using a multivariate linear regression model (aggregate least-squares fit). We used JMP 5.1 (SAS Institute) for all statistical analyses.
Note: Supplementary information is available on the Nature Medicine website.
References
- Seder, R.A. & Hill, A.V. Vaccines against intracellular infections requiring cellular immunity. Nature 406, 793–798 (2000).
Article CAS Google Scholar - Murphy, K.M. & Reiner, S.L. The lineage decisions of helper T cells. Nat. Rev. Immunol. 2, 933–944 (2002).
Article CAS Google Scholar - Bucy, R.P. et al. Heterogeneity of single cell cytokine gene expression in clonal T cell populations. J. Exp. Med. 180, 1251–1262 (1994).
Article CAS Google Scholar - Openshaw, P. et al. Heterogeneity of intracellular cytokine synthesis at the single-cell level in polarized T helper 1 and T helper 2 populations. J. Exp. Med. 182, 1357–1367 (1995).
Article CAS Google Scholar - Roman, E. et al. CD4 effector T cell subsets in the response to influenza: heterogeneity, migration, and function. J. Exp. Med. 196, 957–968 (2002).
Article CAS Google Scholar - Wu, C.Y. et al. Distinct lineages of TH1 cells have differential capacities for memory cell generation in vivo. Nat. Immunol. 3, 852–858 (2002).
Article CAS Google Scholar - Hayashi, N., Liu, D., Min, B., Ben-Sasson, S.Z. & Paul, W.E. Antigen challenge leads to in vivo activation and elimination of highly polarized TH1 memory T cells. Proc. Natl. Acad. Sci. USA 99, 6187–6191 (2002).
Article CAS Google Scholar - Younes, S.A. et al. HIV-1 viremia prevents the establishment of interleukin 2-producing HIV-specific memory CD4+ T cells endowed with proliferative capacity. J. Exp. Med. 198, 1909–1922 (2003).
Article CAS Google Scholar - Reece, W.H. et al. A CD4(+) T-cell immune response to a conserved epitope in the circumsporozoite protein correlates with protection from natural Plasmodium falciparum infection and disease. Nat. Med. 10, 406–410 (2004).
Article CAS Google Scholar - Cooper, A.M. et al. Disseminated tuberculosis in interferon γ gene-disrupted mice. J. Exp. Med. 178, 2243–2247 (1993).
Article CAS Google Scholar - Flynn, J.L. et al. An essential role for interferon γ in resistance to Mycobacterium tuberculosis infection. J. Exp. Med. 178, 2249–2254 (1993).
Article CAS Google Scholar - Wang, Z.E., Reiner, S.L., Zheng, S., Dalton, D.K. & Locksley, R.M. CD4+ effector cells default to the Th2 pathway in interferon γ-deficient mice infected with Leishmania major. J. Exp. Med. 179, 1367–1371 (1994).
Article CAS Google Scholar - Elias, D., Akuffo, H. & Britton, S. PPD induced in vitro interferon gamma production is not a reliable correlate of protection against Mycobacterium tuberculosis. Trans. R. Soc. Trop. Med. Hyg. 99, 363–368 (2005).
Article CAS Google Scholar - Gicheru, M.M. et al. Vervet monkeys vaccinated with killed Leishmania major parasites and interleukin-12 develop a type 1 immune response but are not protected against challenge infection. Infect. Immun. 69, 245–251 (2001).
Article CAS Google Scholar - Leal, I.S., Smedegard, B., Andersen, P. & Appelberg, R. Failure to induce enhanced protection against tuberculosis by increasing T-cell-dependent interferon-γ generation. Immunology 104, 157–161 (2001).
Article CAS Google Scholar - Oliveira, M.R. et al. Germ-free mice produce high levels of interferon-gamma in response to infection with Leishmania major but fail to heal lesions. Parasitology 131, 477–488 (2005).
Article CAS Google Scholar - Blackwell, J.M. Genetic susceptibility to leishmanial infections: studies in mice and man. Parasitology 112 (suppl.), S67–S74 (1996).
PubMed Google Scholar - Bloom, B.R., Flynn, J., McDonough, K., Kress, Y. & Chan, J. Experimental approaches to mechanisms of protection and pathogenesis in M. tuberculosis infection. Immunobiology 191, 526–536 (1994).
Article CAS Google Scholar - Bogdan, C., Moll, H., Solbach, W. & Rollinghoff, M. Tumor necrosis factor-alpha in combination with interferon-gamma, but not with interleukin 4 activates murine macrophages for elimination of Leishmania major amastigotes. Eur. J. Immunol. 20, 1131–1135 (1990).
Article CAS Google Scholar - Liew, F.Y., Li, Y. & Millott, S. Tumor necrosis factor-alpha synergizes with IFN-gamma in mediating killing of Leishmania major through the induction of nitric oxide. J. Immunol. 145, 4306–4310 (1990).
CAS PubMed Google Scholar - Perfetto, S.P., Chattopadhyay, P.K. & Roederer, M. Seventeen-colour flow cytometry: unravelling the immune system. Nat. Rev. Immunol. 4, 648–655 (2004).
Article CAS Google Scholar - Scott, P. Development and regulation of cell-mediated immunity in experimental leishmaniasis. Immunol. Res. 27, 489–498 (2003).
Article CAS Google Scholar - Belkaid, Y., Piccirillo, C.A., Mendez, S., Shevach, E.M. & Sacks, D.L. CD4+CD25+ regulatory T cells control Leishmania major persistence and immunity. Nature 420, 502–507 (2002).
Article CAS Google Scholar - Uzonna, J.E., Wei, G., Yurkowski, D. & Bretscher, P. Immune elimination of Leishmania major in mice: implications for immune memory, vaccination, and reactivation disease. J. Immunol. 167, 6967–6974 (2001).
Article CAS Google Scholar - Modabber, F. Vaccines against leishmaniasis. Ann. Trop. Med. Parasitol. 89 (suppl 1), 83–88 (1995).
Article CAS Google Scholar - Rhee, E.G. et al. Vaccination with heat-killed leishmania antigen or recombinant leishmanial protein and CpG oligodeoxynucleotides induces long-term memory CD4+ and CD8+ T cell responses and protection against Leishmania major infection. J. Exp. Med. 195, 1565–1573 (2002).
Article CAS Google Scholar - von Stebut, E. et al. Skin-derived macrophages from _Leishmania major_-susceptible mice exhibit interleukin-12- and interferon-γ-independent nitric oxide production and parasite killing after treatment with immunostimulatory DNA. J. Invest. Dermatol. 119, 621–628 (2002).
Article CAS Google Scholar - Barouch, D.H. Rational design of gene-based vaccines. J. Pathol. 208, 283–289 (2006).
Article CAS Google Scholar - Tatsis, N. & Ertl, H.C. Adenoviruses as vaccine vectors. Mol. Ther. 10, 616–629 (2004).
Article CAS Google Scholar - Shiver, J.W. et al. Replication-incompetent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 415, 331–335 (2002).
Article CAS Google Scholar - Heppner, D.G., Jr et al. Towards an RTS,S-based, multi-stage, multi-antigen vaccine against falciparum malaria: progress at the Walter Reed Army Institute of Research. Vaccine 23, 2243–2250 (2005).
Article CAS Google Scholar - Wang, J. et al. Single mucosal, but not parenteral, immunization with recombinant adenoviral-based vaccine provides potent protection from pulmonary tuberculosis. J. Immunol. 173, 6357–6365 (2004).
Article CAS Google Scholar - Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Article CAS Google Scholar - Andersen, P. & Doherty, T.M. The success and failure of BCG — implications for a novel tuberculosis vaccine. Nat. Rev. Microbiol. 3, 656–662 (2005).
Article CAS Google Scholar - Zaph, C., Uzonna, J., Beverley, S.M. & Scott, P. Central memory T cells mediate long-term immunity to Leishmania major in the absence of persistent parasites. Nat. Med. 10, 1104–1110 (2004).
Article CAS Google Scholar - Wherry, E.J., Blattman, J.N., Murali-Krishna, K., van der Most, R. & Ahmed, R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77, 4911–4927 (2003).
Article CAS Google Scholar - Constant, S.L. & Bottomly, K. Induction of Th1 and Th2 CD4+ T cell responses: the alternative approaches. Annu. Rev. Immunol. 15, 297–322 (1997).
Article CAS Google Scholar - Jelley-Gibbs, D.M. et al. Repeated stimulation of CD4 effector T cells can limit their protective function. J. Exp. Med. 201, 1101–1112 (2005).
Article CAS Google Scholar - Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
Article CAS Google Scholar - Jelley-Gibbs, D.M. et al. Unexpected prolonged presentation of influenza antigens promotes CD4 T cell memory generation. J. Exp. Med. 202, 697–706 (2005).
Article CAS Google Scholar - Steinman, R.M. & Hemmi, H. Dendritic cells: translating innate to adaptive immunity. Curr. Top. Microbiol. Immunol. 311, 17–58 (2006).
CAS PubMed Google Scholar - O'Garra, A. Cytokines induce the development of functionally heterogeneous T helper cell subsets. Immunity 8, 275–283 (1998).
Article CAS Google Scholar - Oxenius, A., Zinkernagel, R.M. & Hengartner, H. Comparison of activation versus induction of unresponsiveness of virus-specific CD4+ and CD8+ T cells upon acute versus persistent viral infection. Immunity 9, 449–457 (1998).
Article CAS Google Scholar - Wille-Reece, U. et al. Toll-like receptor agonists influence the magnitude and quality of memory T cell responses after prime-boost immunization in nonhuman primates. J. Exp. Med. 203, 1249–1258 (2006).
Article CAS Google Scholar - Sacks, D. & Sher, A. Evasion of innate immunity by parasitic protozoa. Nat. Immunol. 3, 1041–1047 (2002).
Article CAS Google Scholar - Betts, M.R. et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107, 4781–4789 (2006).
Article CAS Google Scholar - Skeiky, Y.A. et al. Protective efficacy of a tandemly linked, multi-subunit recombinant leishmanial vaccine (Leish-111f) formulated in MPL adjuvant. Vaccine 20, 3292–3303 (2002).
Article CAS Google Scholar - Belkaid, Y. et al. A natural model of Leishmania major infection reveals a prolonged “silent” phase of parasite amplification in the skin before the onset of lesion formation and immunity. J. Immunol. 165, 969–977 (2000).
Article CAS Google Scholar - Perfetto, S.P. et al. Amine reactive dyes: an effective tool to discriminate live and dead cells in polychromatic flow cytometry. J. Immunol. Methods 313, 199–208 (2006).
Article CAS Google Scholar
Acknowledgements
TNF-specific antibody (MP6-XT22) was provided by F.D. Finkelman (University of Cincinnati). This research was supported by the Intramural Research Program of the NIH, NIAID.
Author information
Authors and Affiliations
- Cellular Immunology Section, Vaccine Research Center, National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH), 40 Convent Drive, Bethesda, 20892, Maryland, USA
Patricia A Darrah, Dipti T Patel, Paula M De Luca, Ross W B Lindsay, Dylan F Davey, Barbara J Flynn & Robert A Seder - Department of Infectious Disease Immunology, Statens Serum Institute, Artillerivej 5, Copenhagen S, DK-2300, Denmark
Søren T Hoff & Peter Andersen - Infectious Disease Research Institute, 1124 Columbia Street, Seattle, 98104, Washington, USA
Steven G Reed - Laboratory of Mycobacterial Diseases and Cellular Immunology, Center for Biologics Evaluation and Research, Food and Drug Administration, 29 Lincoln Drive, Bethesda, 20892, MD, USA
Sheldon L Morris - ImmunoTechnology Section, Vaccine Research Center, NIAID, NIH, 40 Convent Drive, Bethesda, 20892, Maryland, USA
Mario Roederer
Authors
- Patricia A Darrah
You can also search for this author inPubMed Google Scholar - Dipti T Patel
You can also search for this author inPubMed Google Scholar - Paula M De Luca
You can also search for this author inPubMed Google Scholar - Ross W B Lindsay
You can also search for this author inPubMed Google Scholar - Dylan F Davey
You can also search for this author inPubMed Google Scholar - Barbara J Flynn
You can also search for this author inPubMed Google Scholar - Søren T Hoff
You can also search for this author inPubMed Google Scholar - Peter Andersen
You can also search for this author inPubMed Google Scholar - Steven G Reed
You can also search for this author inPubMed Google Scholar - Sheldon L Morris
You can also search for this author inPubMed Google Scholar - Mario Roederer
You can also search for this author inPubMed Google Scholar - Robert A Seder
You can also search for this author inPubMed Google Scholar
Contributions
P.A.D. designed experiments; developed the multiparameter flow cytometry panel for mice with D.T.P. and M.R.; performed animal studies, intracellular cytokine staining and ELISAs; analyzed and interpreted data; generated figures; and wrote the manuscript with R.A.S. D.T.P assisted in development of the flow cytometry panel, animal studies, intracellular cytokine staining, ELISAs and data analysis. P.M.D.L., R.W.B.L. and D.F.D. assisted in animal studies. B.J.F. performed ELISPOT assays. P.A. provided samples from BCG-vaccinated humans and S.T.H. analyzed these samples with P.A.D. and M.R. S.G.R. provided MML antigen and MML-ADV. S.L.M. provided BCG-vaccinated mice. M.R. assisted in development of the multiparameter flow cytometry panel, data and statistical analysis as well as in preparation of figures and editing of the manuscript. R.A.S. supervised this project, designed experiments, interpreted data and wrote the manuscript with P.A.D.
Corresponding author
Correspondence toRobert A Seder.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Epitope mapping and assessment of inhibitory cytokines following vaccination. (PDF 489 kb)
Supplementary Fig. 2
Functional characterization of distinct populations of Th1 responses using multiparameter flow cytometry (PDF 489 kb)
Supplementary Fig. 3
Magnitude and quality of TH1 responses in lymphoid and non-lymphoid organs after vaccination. (PDF 489 kb)
Supplementary Fig. 4
Proliferative capacity of CD4+ T cells after vaccination. (PDF 489 kb)
Supplementary Fig. 5
Functional hierarchy of TH1 cytokine production in lymphoid and non-lymphoid organs after vaccination. (PDF 489 kb)
Supplementary Fig. 6
Protection in mice after vaccination with high-dose MML-ADV when challenged at the peak of the TH1 response. (PDF 489 kb)
Supplementary Fig. 7
Kinetics and quality of the TH1 influence the degree of protection after infection. (PDF 489 kb)
Supplementary Fig. 8
Durability of TH1 responses after immunization. (PDF 489 kb)
Rights and permissions
About this article
Cite this article
Darrah, P., Patel, D., De Luca, P. et al. Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major.Nat Med 13, 843–850 (2007). https://doi.org/10.1038/nm1592
- Received: 22 February 2007
- Accepted: 17 April 2007
- Published: 10 June 2007
- Issue Date: July 2007
- DOI: https://doi.org/10.1038/nm1592