Mechanical signaling through the cytoskeleton regulates cell proliferation by coordinated focal adhesion and Rho GTPase signaling (original) (raw)
Abstract
The notion that cell shape and spreading can regulate cell proliferation has evolved over several years, but only recently has this been linked to forces from within and upon the cell. This emerging area of mechanical signaling is proving to be wide-spread and important for all cell types. The microenvironment that surrounds cells provides a complex spectrum of different, simultaneously active, biochemical, structural and mechanical stimuli. In this milieu, cells probe the stiffness of their microenvironment by pulling on the extracellular matrix (ECM) and/or adjacent cells. This process is dependent on transcellular cell–ECM or cell–cell adhesions, as well as cell contractility mediated by Rho GTPases, to provide a functional linkage through which forces are transmitted through the cytoskeleton by intracellular force-generating proteins. This Commentary covers recent advances in the underlying mechanisms that control cell proliferation by mechanical signaling, with an emphasis on the role of 3D microenvironments and in vivo extracellular matrices. Moreover, as there is much recent interest in the tumor–stromal interaction, we will pay particular attention to exciting new data describing the role of mechanical signaling in the progression of breast cancer.
Key words: Cancer, Cell proliferation, Cytoskeleton, Focal adhesion, Mechanotransduction, Rho GTPAse
Introduction
In the complex extracellular matrix (ECM) microenvironment cells encounter a multitude of coordinated, simultaneously active stimuli that are biochemical, structural and mechanical in nature (Fig. 1A). In this context, mechanical forces have a role in regulating the development and correct function of virtually every tissue in the human body. In fact, the local mechanical interaction between the cell and its microenvironment is emerging as a unifying principle that connects micro- and macro-scale tissue architecture with cell shape, organization and differentiation (for reviews, see Discher et al., 2005; Vogel and Sheetz, 2006; Wang, N. et al., 2009; Wozniak and Chen, 2009). For instance, cell and ECM mechanics (Box 1) are known to be motivating factors in cell differentiation (Engler et al., 2008; Engler et al., 2006), migration (Gardel et al., 2008; Hadjipanayi et al., 2009; Lo et al., 2000), morphogenesis (Paszek et al., 2005; Wozniak et al., 2003) and proliferation (Provenzano et al., 2009; Ulrich et al., 2009; Wang et al., 2000). Furthermore, mechanics and associated mechanotransduction (the mechanisms by which cells transduce mechanical stimuli into a biochemical response), is known to have a substantial role in numerous human diseases (e.g. Grinnell, 2003; Hahn and Schwartz, 2009; Robling et al., 2006), including tumor formation and progression (Paszek et al., 2005; Provenzano et al., 2009; Ulrich et al., 2009; Wozniak et al., 2003).
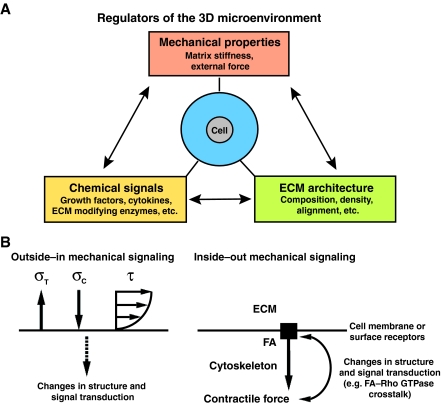
Fig. 1.
The mechanically active 3D microenvironment. (A) The primary factors of the 3D microenvironment that influence cell behavior. A dynamic dependence exists between all three factors, but here the focus is on mechanical signals to and from the ECM and the role these stimuli have in cell proliferation. (B) Outside–in (left) and inside–out (right) mechanical signals. During normal physiological function, cells and tissues in the body experience multi-axial loading that result from a complex superposition of external forces to produce stress in the cell. For example, tensile stress (σT), compressive stress (σC) and shear stress (τ; depicted as the result of fluid flow over the cell) are commonly applied to cells during normal physiological tissue function. Of course, for each of these stimuli there is an equal force that exists in the cell (not shown). Furthermore, during inside–out signaling, chemical energy is converted to mechanical energy in order to generate contractile forces within the cell and to impart stress on the ECM (not shown), which results in an elevated force balance at the FA that influences signal transduction within the cell.
Box 1. Glossary of mechanics terminology
Mechanics
The sub-discipline of physics and engineering devoted to the action of forces and displacements on physical bodies.
Force and stress
Force is a vector (it has magnitude and direction) that produces acceleration of a body in the direction of the applied force. When the resultant force that acts on a body is zero the body is in equilibrium. Stress (σ) is the force per unit area and is a measure of the forces acting between particles in a deformable body.
Deformation and strain
In solid continuum mechanics (i.e. the study of mechanical behavior of materials modeled as a continuous mass rather discrete particles), deformation is an alteration in the shape or size of a body as the result of an applied force. Strain (ε) is a unitless normalization of deformation and for small strains can be described as ε=(L–L0)/L0, where L is the final length of the body and L0 is the initial length (known as ‘engineering’ or ‘Cauchy’ strain). For larger strains (>5–10% strain) as commonly seen in biologic materials, the use of ‘Green’ strain is appropriate: ε=1/2*[(L/L0)2−1].
Stiffness and elastic modulus
Stiffness of an elastic material is the internal resistance to deformation produced by the application of force. It is a structural property (also known as an extensive property) because it depends on the size, organization and shape of the material. By contrast, the elastic modulus is a material property (also known as an intensive property) as it is a normalized metric that is independent of geometric considerations. The elastic modulus (E) describes the tendency of a material to undergo elastic strain when experiencing stress; e.g. the tensile (or Young's) modulus is defined as E=σ/ε (stress/strain) and can be determined by the slope of the stress–strain curve. Compliance, a term commonly used in cell biology, is the inverse of stiffness and is a measure of the softness as opposed to the stiffness of a material.
Contractile and traction force
A cell converts chemical energy to mechanical energy during the process of contraction. Contractile force refers to the force generated by a cell in a 3D environment and is resisted by the stiffness of microenvironment. Because cell-generated contractile force pulls on the ECM, cells stretch the ECM. Therefore, tensile structural and material properties are the most appropriate measures to understand the mechanical interaction between cells and the microenvironment. Traction force is commonly used to describe force generated by a contracting cell on a 2D substrate. Because the cell sits on top of the substrate, the primary force is in-plane with the area experiencing the force, i.e. shear force. Resistance to traction force by the substrate is, therefore, described by the materials resistance to shear stress, known as the shear modulus or modulus of rigidity.
Viscoelasticity
Biologic materials display both elastic and viscoelastic behavior. Viscoelastic materials display both elastic (solid) and viscous (fluid) characteristics. Whereas elastic materials store energy under deformation, viscoelastic materials dissipate energy (e.g. the gel cushion in running shoes). In addition, a main feature of viscoelastic materials is the presence of a time-dependent relationship between stress and strain. This time dependence is commonly described by two main characteristics of viscoelastic materials: creep (the time-dependent change in strain under constant stress) and stress relaxation (the decrease in stress over time when material strain is constant). Interestingly, in most biologic materials the most significant creep or stress relaxation occurs very rapidly (often on the order of seconds) and then the strain or stress, respectively, approaches steady-state levels. As such, owing to the dynamic nature of tensional homeostasis over a long period of time (relative to the time scale for which many viscoelastic materials approach steady-state value for creep and relaxation), the influence of viscoelastic behavior in the microenvironment is currently not well-understood and an increased understanding of viscoelastic behavior in the microenvironment is thus likely to provide additional insight into mechanotransduction.
Mechanical stress and associated cell strain, can arise from externally imposed, ‘outside–in’ mechanical stimuli (Fig. 1B), which is common during force transmission to cells via the ECM in load-bearing tissues such as bone (Robling et al., 2006). Alternatively, or in coordination with outside–in stimuli, anchored cells also probe their microenvironment to sense and respond to the stiffness of the microenvironment by pulling on the ECM, i.e. ‘inside–out’ mechanical stimuli. Such processes are dependent on ECM adhesions that act as a bridge to transmit force between the ECM and the cellular cytoskeleton, in which myosin-based contractility acts as a primary regulator of cellular traction (contractile) forces (Geiger et al., 2001). Indeed, these intracellular forces have a role in regulating signaling pathways that are involved in fundamental cell processes that determine cell phenotype. For instance, myosin-based contractility has a role in regulating stem cell differentiation (Engler et al., 2006), epithelial morphogenesis (Alcaraz et al., 2008; Gehler et al., 2009; Sahai and Marshall, 2002; Wozniak et al., 2003), branching morphogenesis of epithelial and endothelial cells (Fischer et al., 2009; Moore et al., 2005), and the cancer-associated invasive phenotype that is induced in mammary epithelial cells by stiff 3D matrices (Provenzano et al., 2009; Provenzano et al., 2008b).
Importantly, cells possess the ability to modulate their force generation in response to local stiffness of 2D substrates or 3D microenvironments. Increasing substrate stiffness results in increased traction force in fibroblasts (Lo et al., 2000; Paszek et al., 2005) and epithelial cells (Saez et al., 2005). Moreover, fibroblasts adjust their internal cell stiffness to match substrate stiffness (Solon et al., 2007). Thus, feedback loops exist by which cells sense the stiffness of the microenvironment and exert contractile force at a magnitude that scales with this stiffness (Fig. 2), a characteristic that is a crucial determinant of cell behavior. If the stiffness of the microenvironment is abnormally low or there is no substrate, cells will experience anchorage-independent conditions under which normally adherent non-transformed cells will not survive (Ma et al., 2008). However, under normal physiological conditions, a dynamic force balance – termed tensional homeostasis – occurs between the cell and the microenvironment, and the cell will proliferate at normal levels (Klein et al., 2009). By contrast, if matrix stiffness is uncharacteristically elevated, an abnormally elevated tensional homeostasis arises that can result in aberrant cellular behaviors. For example, increased matrix stiffness associated with the increased deposition, altered composition and alignment of the collagenous stroma that accompanies breast tumor progression can promote disruption of epithelial architecture (Paszek et al., 2005; Provenzano et al., 2008a; Wozniak et al., 2003). Cells in this environment hyperactivate a mechanically regulated signaling loop that results in increased expression of a conserved set of carcinoma-associated proliferation genes, such as genes that encode cyclins, aurora kinases, cell division cycle regulators and E2F transcription factors, which are important for cell proliferation (Provenzano et al., 2009). Hence, in addition to the cellular microenvironment being an intricate system that includes a complex chemical milieu surrounding the cell, it is now clear that the stiffness of the ECM and the corresponding mechanical response of the cell have a role in regulating fundamental processes – including cell proliferation.
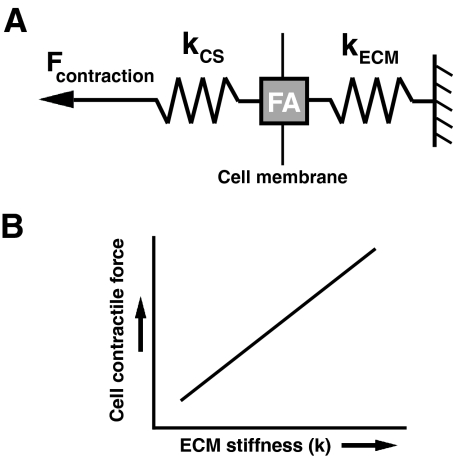
Fig. 2.
Cell contraction force as a function of ECM stiffness. (A) Contractile force is transmitted within the actin cytoskeleton through the FA to the ECM (kCS and kECM represent the stiffness of the actin cytoskeleton and the ECM, respectively). (B) The magnitude of cell-generated contractile force is dependent upon the stiffness of the ECM (kECM). As the stiffness of the microenvironment increases, the magnitude of contractile force also increases in order to maintain tensional homeostasis. Of course, the cellular response to the mechanical properties of the microenvironment is further complicated because of the viscoelastic behavior of the ECM and within the cell itself (not shown; see Box 1). Although the time scale over which cells deform the matrix suggests that the process can be well described by understanding elastic behavior; the physiological implication of viscoelastic phenomena and the degree to which mechanical behavior at cellular scale is dominated by either elastic or viscous effects in a certain situation is not well understood – but is likely to provide additional insight into mechanotransduction.
In this Commentary, we will cover recent advances in our understanding of the regulation of cell proliferation by mechanical signaling. The emphasis is on the role of mechanical signal transduction in the context of 3D microenvironments, which include cell culture models of defined ECM composition and concentration, such as collagen matrices that contain individual cells or cell communities, models of complex ECM mixtures including recombinant basement membrane (Matrigel) and microenvironments that are found within tissues in vivo. We pay particular attention to exciting new data describing the role of mechanical signaling in cancer progression. In this area, the largest volume of work focuses on breast cancer, which can be used as a beacon for understanding other cancers because many of the signaling mechanisms might be similar.
For years, there was the suspicion that cell shape and spreading regulates cell proliferation, but only recently have forces from within and upon the cell been directly linked to these processes. Thus, we will discuss mechanisms by which stiffness of the ECM influences the cell–matrix interaction and focal adhesions, as well as Rho GTPase signaling to transmit mechanical forces through the cytoskeleton, that lead to changes in cell proliferation. Although herein the emphasis is on cancer cell proliferation, the topic is of broad relevance because the mechanisms involved are likely to be at play in multiple cell types during developmental processes (e.g. stem cell differentiation, cellular morphogenesis and generation of tissue architecture) as well as in several proliferative pathologies (e.g. vascular disease, wound repair, lung and liver fibrosis, etc.) that are accompanied by changes in the ECM.
Linking the actin cytoskeleton to the microenvironment – focal adhesions as regulators of mechanical signaling and cell proliferation
Cells form a functional force linkage between intracellular, contractile, force-generating motor proteins, the cytoskeleton and transcellular adhesions (e.g. integrins) to transmit force to the ECM. In addition, cell contacts between neighboring cells via cadherins are linked to the actin cytoskeleton and are also subjected to, and respond to, forces between cells. The subject of cell–cell mechanical signaling is not covered in detail here; force transmission between cells has been reviewed elsewhere (Chen et al., 2004; Ingber, 2006; Janmey and McCulloch, 2007; Wozniak and Chen, 2009). The most-prominent cell adhesion structure is the focal adhesion (FA), which has been defined for cells on 2D surfaces coated with ECM (Burridge et al., 1988). In 3D environments, specialized 3D-matrix adhesions are observed (Cukierman et al., 2001). The differences between 3D-matrix adhesions and those found in 2D systems might account for many of the differences in cell signaling that are observed in 3D culture (referenced throughout this article). FAs are sites of integrin clustering that form a physical link between the actin cytoskeleton and the ECM (Burridge et al., 1988) to transduce force between the cell and its microenvironment (Geiger et al., 2001). Consistent with the concept that FA signaling increases when a cell faces resistance to intracellular contraction owing to the stiffness of the ECM, force transmitted through the actin cytoskeleton results in increased force at the cell-matrix interface, which further promotes FA assembly (Chrzanowska-Wodnicka and Burridge, 1996; Ridley and Hall, 1992). Furthermore, external force applied to integrins, or exposure to a stiff 2D substrate promotes increased FA size and strength (Choquet et al., 1997; Galbraith et al., 2002; Pelham and Wang, 1997; Sniadecki et al., 2007). For 3D-matrix adhesions, we have shown that disruption of either mechanisms of myosin-based contractility or the actin cytoskeleton itself – which releases intracellular tension – diminishes the number of 3D-matrix adhesions that are formed in response to stiff collagen matrices (Provenzano et al., 2009). Moreover, 3D-matrix adhesions do not readily form in compliant matrices but, rather, in stiff matrices that offer resistance to intracellular contractility (Provenzano et al., 2009; Wozniak et al., 2003). However, it should be noted that the presence of FAs or punctate 3D-matrix adhesions in cells within 3D microenvironments is controversial. For example, a recent study has demonstrated that FA proteins are diffuse in HT-1080 fibrosarcoma cells that are cultured in 3D (Fraley et al., 2010), whereas another study using the same cell line has demonstrated robust β1-integrin-positive 3D-matrix adhesions in 3D (Wolf et al., 2003). As more data emerge, evidence that 3D-matrix adhesions are dependent on ECM stiffness might help in addressing these discrepancies.
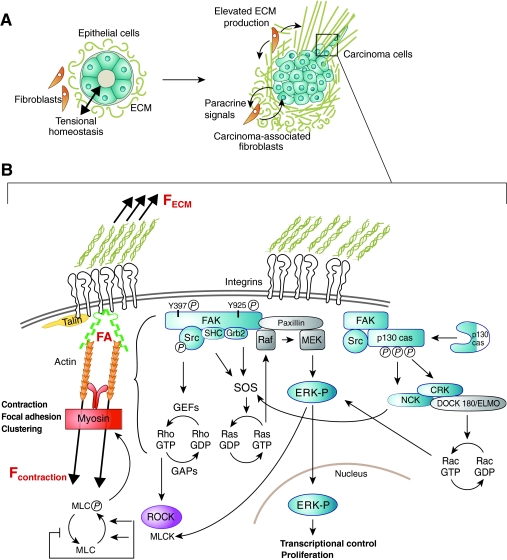
In addition to acting as a physical scaffold, FAs function as a node on which to assemble and regulate a complex signaling network that influences fundamental cell processes including cell proliferation (Mitra et al., 2005; Playford and Schaller, 2004). For instance, FA kinase (FAK), a primary regulator of FA signaling, activates pathways known to promote cell proliferation (Fig. 3). Integrin-stimulated phosphorylation of FAK at tyrosine residue 397 creates a high-affinity site that is recognized by several Src homology 2 (SH2) domain-containing proteins, including Src and Shc (Schaller et al., 1994; Schlaepfer et al., 1998; Xing et al., 1994). Moreover, FAK phosphorylation at tyrosine 925 by Src links FAK to the Ras pathway via growth factor receptor-bound protein 2 (Grb2) (Schlaepfer et al., 1994; Schlaepfer and Hunter, 1996; Schlaepfer et al., 1998). Interestingly, FAK activation has been shown to occur with both outside–in and inside–out mechanical signaling. For example, several studies demonstrate a role for FAK in mechanotransduction by showing that cell or tissue loading affects FAK expression or activation in a number of cell types, such as fibroblasts (Moalli et al., 2001; Molina et al., 2001; Wang, J. G. et al., 2001), osteocytes (Wozniak et al., 2000), chondrocytes (Lee et al., 2000), vascular smooth muscle cells (Li et al., 2009; Nakayama et al., 2003), skeletal muscle cells (Fluck et al., 1999; Gordon et al., 2001) and epithelial cells (Basson et al., 2000; Provenzano et al., 2009; Zhang et al., 2003). FAK is also necessary for mechanically induced osteogenesis (Leucht et al., 2007) and has been implicated in mechanosensing in migrating fibroblasts (Wang, H. B. et al., 2001). FAK is phosphorylated at tyrosine residues 397 and 925 during fibroblast deformation, which promotes phosphorylation of extracellular signal-regulated kinases 1 and 2 (ERK1/2) (Wang, J. G. et al., 2001) and, subsequently, cell proliferation (Wang et al., 2005). Furthermore, mammary epithelial cells in stiff 3D matrices can upregulate FAK phosphorylation (Paszek et al., 2005; Provenzano et al., 2009; Wozniak et al., 2003) and we have shown that an increase in Rho-mediated cell contraction in response to a stiff microenvironment drives the localization of active FAK into 3D-matrix adhesions (Provenzano et al., 2009). It is worth noting that the stiff ECM microenvironment also increases the formation of complexes between FAK and Src and Shc and the mitogen-activated protein kinase (MAPK) pathway member Grb2 – both of which go on to enhance FAK-dependent activation of ERK1/2 (Provenzano et al., 2009) (Fig. 3). Hence, the force balance at the cell-matrix interface, which is established by the contractile response to ECM stiffness, directly regulates classical pathways of proliferation, such as the Ras-MAPK pathway.
Fig. 3.
Inside–out contractile force as a regulator of cell signaling. Stiffness of the extracellular matrix influences the magnitude of the contractile force of the cell that is transmitted to the ECM through integrins. (A) Increased ECM deposition by carcinoma-associated fibroblasts (CAFs) produces a stiff microenvironment for carcinoma cells. The dense ECM contains CAFs that signal to carcinoma cells, provides micro- and/or nano-structures that influence cell behavior, and has a stiffness to which the cells respond. (B) Elevated stiffness of the extracellular matrix results in elevated forces (F) at the cell–matrix interface, and promotes 3D-matrix adhesion formation and maturation that leads to the activation of highly dynamic signaling networks that regulate fundamental cell processes such as proliferation.
Regulation of cell proliferation by FA signaling is, however, more complex than providing merely a direct linkage to the Ras pathway. In addition to this direct activation of pathways that are known to regulate cell proliferation, FA components, such as FAK and Src, also display considerable crosstalk with receptor tyrosine kinases (RTKs) and affect the regulation of their activity (Aplin and Juliano, 1999; Baron et al., 1998; Benlimame et al., 2005; Casamassima and Rozengurt, 1998; Sieg et al., 2000). Examples are the epidermal growth factor (EGF) and insulin-like growth factor (IGF) family of receptors, which are also known to promote cell proliferation (Baselga and Swain, 2009; Pollak, 2008). Although a requirement for autocrine EGF signaling to induce phosphorylation of ERKs in compressed cells has been demonstrated (Tschumperlin et al., 2004), little is currently known regarding the role mechanical signals have in mediating crosstalk between FAs and RTKs.
An additional level of complexity arises from reports that ERK is localized at FAs in complexes that also contain the FA proteins paxillin, Src and FAK [together with indications that paxillin associates with Raf and MAPK kinase (MEK)] to regulate epithelial cell migration and morphogenesis (Ishibe et al., 2004; Ishibe et al., 2003; Liu et al., 2002; Webb et al., 2004). These findings have interesting implications for a possibly direct mechanical regulation of the MAPK pathway to cause cell proliferation, because FAK, Src and paxillin are regulated in response to mechanical signals.
FAK can also directly bind to, and promote Src-mediated phosphorylation of, p130Cas, an adaptor protein that mediates several cellular events associated with cell adhesion and mitogen signaling, and that undergoes stretch-dependent activation that results in its increased phosphorylation by Src family kinases (Sawada et al., 2006). Activation of p130Cas, in turn, scaffolds a complex between Crk, Dock180 and the engulfment and cell motility (ELMO) protein, which serves as a guanine exchange factor (GEF) for Rac. Moreover, p130Cas provides a scaffold for the non-catalytic region of tyrosine kinase adapator protein (Nck), which then activates the Ras–ERK pathway, suggesting that mechanical signals regulate cell proliferation in part through different signaling routes that all lead to activation of ERKs (Fig. 3). Hence, although it remains to be fully elucidated whether MAPK components that are localized to FAs have a role in regulating proliferation, current data suggest that additional links exist between mechanical signals transmitted through the actin cytoskeleton to FAs and the signaling cascades that regulate cell proliferation.
Finally, how mechanical signals are transmitted through the structural components of the cytoskeletal to FAs is likely to have a role in signaling events that regulate cell proliferation. Foremost, mechanical activation of signaling pathways at FAs requires an intact and well-regulated cytoskeleton to effectively transmit force from the contractile machinery to the FA proteins, suggesting that changes in the cytoskeleton structure can differentially influence the mechanical stimuli presented to FAs. It is known that the actin cytoskeleton displays complex elastic and viscoelastic behavior (e.g. Gardel et al., 2006; Liu et al., 2006; Luan et al., 2008; Tseng et al., 2004; Xu et al., 2000). As such, it is possible that an increased understanding of how mechanical signals regulate cytoskeleton remodeling through actin modifying proteins and how temporal behavior of cytoskeleton mechanics modulate differential signal transduction, will improve our understanding of mechanotransduction. For instance, such information might provide insight into whether there are direct mechanisms of force transmission through the cell to the nucleus that influence gene expression (Wang, N. et al., 2009), which in turn could regulate the expression of cell proliferation genes. Thus, although our understanding of the link between cytoskeletal mechanics and mechanotransduction remains incomplete at present, it is clear that force transmission through the cytoskeleton significantly influences FA signaling, and that a complex feedback loop exists between cell proliferation and pathways that regulate intracellular contraction to produce force through the cytoskeleton to regulate FA signaling.
Rho GTPases and the proliferative response to mechanical cues
Cells sense the stiffness of their environment by generating forces from within the cell that pull against the extracellular matrix. These contractile forces are largely based on actin–myosin interactions, because inhibition of actin or myosin activity abolishes the ability of epithelial cells to contract 3D matrices and to undergo tubulogenesis (Wozniak et al., 2003). The small GTPase Rho is a key regulator of intracellular contractility and, thus, allows cells to sense matrix stiffness and respond to mechanical cues. This function is largely exerted through the Rho effector protein Rho-associated protein kinase (ROCK) because epithelial cells with modified ROCK or Rho activity – i.e. with levels outside of the physiological range required for appropriate morphogenesis – no longer respond effectively to matrix stiffness (Paszek et al., 2005; Provenzano et al., 2009; Wozniak et al., 2003). As a result of Rho- or ROCK-mediated contractility, cells in compliant matrices are able to reorganize the matrix and assume a more-rounded morphology. Interestingly, cell shape has long been linked to regulated proliferation (Chen et al., 1997; Folkman and Moscona, 1978; Huang et al., 1998), although the underlying mechanism is not well-understood. Data suggest that cell shape has a dominant role in regulating cell proliferation: when cells are placed on defined 2D surfaces in which their shape is confined, they have restricted proliferation (Chen et al., 1997). Thus, an important mechanism by which matrix compliance regulates proliferation is likely to be the effect it has on cell shape.
Several cell types cultured in a compliant environment show diminished proliferation and increased differentiation compared with cells in stiff environments (Fringer and Grinnell, 2003; Grinnell et al., 2003; Helm et al., 2007; Koyama et al., 1996; Ng et al., 2005; Wang et al., 2000; Wozniak et al., 2003). Stiff matrices, in turn, lead to increased Rho activation (Bhadriraju et al., 2007; Provenzano et al., 2009; Wozniak et al., 2003). The exact mechanism by which this occurs is still being elucidated. Several Rho GEFs are regulated by phosphorylation and, therefore, might be targets of increased integrin-mediated signaling through FAK and ERK1/2 that, because of increased mechanical tension, occurs in stiff environments (Provenzano et al., 2009; Rosenfeldt and Grinnell, 2000). Some of these pathways are discussed below.
Rho has been linked to increased cell proliferation in many cancers, in which it is increased in both amount and activation state (Faried et al., 2007; Sterpetti et al., 2006; Wang, H. B. et al., 2009; Zhang et al., 2009). Mammary carcinoma cells proliferate in response to stiff matrices, and this was shown to be mediated in part by Rho (Paszek et al., 2005; Provenzano et al., 2009; Wozniak et al., 2003). In addition to cancer, a link between Rho activation, mechanical signaling and proliferation has also been demonstrated in other cell types. For example, vascular smooth muscle cells in blood vessels proliferate in a Rho-dependent manner in response to deformation (Halka et al., 2008; Qi et al., 2010). Similarly, skeletal muscle cells proliferate following cyclic mechanical strain, and this involves regulation of FAK, NFκB and the Rho GTPase Rac1 (Kumar et al., 2004). The interplay between Rho and FAK is complex, as non-phosphorylated FAK inhibits Rho-mediated cellular proliferation in endothelial cells (Pirone et al., 2006). Once Rho is activated in response to stiff matrices or by mechanical strain, there are several pathways by which it may signal in order to stimulate proliferation.
The Rho family of GTPases is involved in cell-cycle progression at several points. Activation of the family members Rho, Rac and Cdc42 can lead to expression of cyclins D and E (for reviews, see Assoian and Klein, 2008; Pruitt and Der, 2001). They are also involved in timing the activity of cyclin D1, because Rho activity switches off the early activation of cyclin D1 by Rac and Cdc42 and, instead, sustains ERK1/2 activation, which ensures subsequent cyclin D1 expression in the mid-G1 phase of the cell cycle (Welsh et al., 2001). Rho GTPases also activate p38 mitogen-activated protein kinase and Jun N-terminal kinase (JNK) through the MAPK kinase – mixed-lineage kinase (MKK–MLK) pathway, which results in the transcriptional regulation of genes associated with cell proliferation (Minden et al., 1995; Neisch et al., 2010; Philips et al., 2000; Whitehead et al., 1999). In addition, Rho family GTPases activate serum response factor (SRF), which can enhance proliferation through increased transcription of cyclins (Hill et al., 1995). Transcription mediated by SRF is regulated in a competitive manner by either the ternary complex factor (TCF) or the myocardin-related transcription factor (MRTF), depending on the signaling inputs (Gineitis and Treisman, 2001; Wang et al., 2004). MRTF is particularly relevant to mechanical regulation of transcription and cellular proliferation because it is inhibited by G-actin; thus, the effect of Rho GTPases on stimulating actin polymerization allows MRTF to enter the nucleus and regulate SRF (reviewed in Olson and Nordheim, 2010). In addition to proliferation, MRTF and SRF contribute to metastasis of both breast carcinoma and melanoma cells (Medjkane et al., 2009). Here, a role of cellular contractility depending on actin and myosin is suggested by the finding that one of the targets of MRTF and SRF, myosin 9, is also required for metastasis (Medjkane et al., 2009). A further level of regulation occurs through suppressor of cancer cell invasion 1 (SCA1), which can form a ternary complex with SRF to inhibit its target proteins. In this case, a key target is β1-integrin, which is upregulated upon loss of SCA1 and induces invasion of breast carcinoma cells (Brandt et al., 2009). A number of MRTF and SRF targets are also found in skeletal and smooth muscle cells, and here Rho signaling to SRF induces both proliferation and expression of differentiation-specific genes that are involved in myogenesis (Gopinath et al., 2007; Kuwahara et al., 2005; Lockman et al., 2004).
Rho GTPases also signal to phosphoinositide 3-kinase (PI 3-kinase), which activates proliferation through the Akt pathway. For example, both RhoA and RhoC are involved in promoting proliferation in gastric carcinoma that depends on PI 3-kinase and Akt (Sun et al., 2007). Of relevance to mechanical regulation of cell proliferation is the observation that cells cannot progress through G1 into S phase in the absence of integrin-mediated adhesion, which appears to rely on Rho GTPases. Rho activation is necessary to remove the inhibition of cell-cycle progression that is imposed by p21 and p27 (Zhang et al., 2009). Thus, enhanced integrin-mediated signaling that occurs when cells are in stiff matrices will activate Rho GTPases and promote progression through G1 by induction of cell-cycle stimulators and loss of cell-cycle inhibitors.
Rho GTPases also regulate several of the steps involved in mitosis (for a review, see Schlessinger et al., 2009). Rho, in particular, is involved in initial cell rounding that heralds the start of mitosis and, later, in the development of the cleavage furrow that leads to cytokinesis (Glotzer, 2005). Two Rho-specific GEFs, epithelial cell transforming sequence 2 (Ect2) and GEF-H1, are involved in the precise regulation of Rho during cytokinesis. Ect2 is recruited to the central spindle through the action of Polo-like kinase 1 (Burkard et al., 2007; Petronczki et al., 2007), which also regulates the positioning of RhoA during cytokinesis (Burkard et al., 2007), whereas the Rho-specific GTPase activating protein (GAP) p190RhoGAP is degraded during cleavage-furrow formation in order to sustain mitosis (Su et al., 2009). Thus, an intriguing possibility – although there are currently no data to support or refute this idea – is that increased cell proliferation within stiff matrices is, in part, due to an upregulation of Rho pathways that are also necessary for mitosis. As such, it is important to note that GEF-H1 is activated by mechanical strain in vascular endothelial cells (Birukova et al., 2010), thus, suggesting it has a direct role in mediating cell proliferation in response to mechanical signals.
In polarized epithelial cells, GEF-H1 is kept inactive by being sequestered to tight junctions (TJs). There, it binds to cingulin, a TJ protein whose function is not well understood and, thus, might contribute to contact inhibition of cell proliferation (Aijaz et al., 2005; Citi et al., 2009; Nie et al., 2009). Release of GEF-H1 from tight junctions, for example during epithelial–mesenchymal transition (EMT), activates Rho, which then – to promote proliferation – collaborates with ZO-1 associated nucleic-acid binding protein (ZONAB), a transcription factor that increases expression of cyclin D1 (Nie et al., 2009). In addition to GEFs, RhoGAPs are also a mechanism through which mechanical signaling can regulate cell proliferation. For instance, p190RhoGAP is mechanosensitive (Mammoto et al., 2009) and is regulated during cell adhesion (Arthur and Burridge, 2001; Arthur et al., 2000). p190RhoGAP is phosphorylated by Src (Arthur et al., 2000; Parsons and Parsons, 2004), which allows it to be regulated downstream of both integrin and growth factor pathways (Parsons and Parsons, 2004). Furthermore, both vascular endothelial growth factor (VEGF) and matrix elasticity can control p190RhoGAP in order to regulate two antagonistic transcriptional factors (TFII-I and GATA2) that control angiogenic factors (Mammoto et al., 2009), suggesting that RhoGAPs and GEFs are potential convergence points, at which the microenvironment and soluble factors coordinate cell responses.
Rho GTPases also have a role in inhibiting apoptosis and, through this function, might also contribute to cancer progression. Rho–ROCK and Cdc42–Rac–PAK signaling networks are required to regulate Bcl2 family members and inhibit activation of caspase 3 in gastric carcinoma cells (He et al., 2008). Furthermore, Rho activation in hepatocellular carcinoma results in upregulation of the anti-apoptotic Bcl2 and increased phosphorylation of the pro-apoptotic Bcl2-associated death promoter (BAD), thus reducing apoptosis and resulting in cells that are resistant to genotoxic stress (Sterpetti et al., 2006). Interestingly, mammary epithelial cells downregulate several pro-apoptotic genes when they are cultured in a stiff matrix (Provenzano et al., 2009), further supporting the idea that mechanical stimuli promote increased cell numbers through both increased proliferation and resistance to apoptosis.
Regulation of stem cell fate, as well as maintenance of the self-renewing population of stem cells, are nuances of cell proliferation that are also regulated by Rho. Self-renewal is maintained in part through Wnt signaling. A growing body of work suggests that both the canonical and non-canonical Wnt pathways are partly regulated by Rho (reviewed in Schlessinger et al., 2009). The commitment of cells that differentiate to a particular lineage is also regulated by cytoskeletal tension and Rho. In a manner that is also linked to cell shape, mesenchymal stem cells (MSCs) adopt an osteogenic fate when RhoA is activated and an adipocyte fate when RhoA is inhibited (McBeath et al., 2004). Furthermore, MSCs cultured on substrates of a different stiffness will commit to a lineage whose microenvironment corresponds to the substrate stiffness, i.e. to osteogenic lineages on stiffer substrates with an elastic modulus approximating that of embryonic bone microenvironments, and to neurogenic lineages on softer substrates that have an elastic modulus similar to that of neural tissue (Engler et al., 2006).
Mechanical regulation of cell proliferation in cancer
The increased density and altered composition of the fibroblast-derived ECM, and its influence on carcinoma cells during tumor progression (Amatangelo et al., 2005; Cukierman and Bassi, 2010; Serebriiskii et al., 2008) suggests a role for the physical properties of the ECM in tumor progression. In addition, reports by Grinnell and co-workers demonstrated that reducing resistance to cell contractile force in fibroblasts cultured in 3D collagen matrices inhibits cell proliferation by reducing phosphorylation of ERKs, downregulation of cyclin D1 and increasing levels of the cell-cycle inhibitor cyclin-dependent kinase inhibitor 1B (CDKN1B, also known as Kip1) (Fringer and Grinnell, 2001; Rosenfeldt and Grinnell, 2000). Extending these studies in breast carcinoma cells, Wozniak and colleagues demonstrated a link between cellular contractility and proliferation (Wozniak et al., 2003). Under conditions of high resistance to cell contraction by the 3D matrix, proliferation is high, whereas reduced resistance to cell contraction decreases the proliferation of carcinoma cells (Wozniak et al., 2003). Consistent with these findings, normal and transformed mammary epithelial cells cultured in 3D matrices of increasing stiffness result in increasingly larger colonies (Paszek et al., 2005) and a more proliferative population of cells (Provenzano et al., 2009; Provenzano et al., 2008a). Indeed, culturing mammary epithelial cells in matrices with high stiffness increases the expression of genes that have been identified as the breast carcinoma ‘proliferation signature’ (Whitfield et al., 2006), which negatively correlates with patient survival as shown by us and others (Provenzano et al., 2009; Whitfield et al., 2006). Expression of this proliferation signature is dependent on ERKs, and also serves to induce an invasive phenotype (Provenzano et al., 2009). Furthermore, in addition to transcripts associated with the G1 phase of the cell cycle, there is an upregulation of transcripts that regulate G2 and G2–M transition and that display a strong statistical enrichment for computationally predicted transcription factor binding sites for, and hence regulation by, the transcription factor p53. The tumor suppressor protein p53 is known to be deleted or mutated in numerous human cancers (Junttila and Evan, 2009) and functions as a regulator of G1–S and G2–M transitions (Taylor and Stark, 2001). Intriguingly, FAK binds directly to p53, which results in reduced transcriptional activity of p53 (Golubovskaya et al., 2005) and its inactivation through murine double minute 2 (Mdm2)-dependent p53 ubiquitylation, whereas loss of FAK results in increased amounts of active p53 that impair cell proliferation (Lim et al., 2008). Taken together, these studies suggest that mechanical signals that active FA proteins including FAK emerge as direct mediators of proteins known to be important in cancer – such as p53.
In addition to findings in epithelial carcinoma, recent work has shown that cell lines derived from glioma also respond to substrate stiffness (Ulrich et al., 2009). Here, increasing rigidity of fibronectin-coated substrates substantially increase glioma cell proliferation, suggesting a role for abnormally stiff microenvironments in promoting brain tumor progression. Interestingly, the authors showed that inhibition of ROCK- and myosin-based cellular contractility impede the stiffness-induced actin architecture of the cytoskeleton and the associated spread-cell phenotype (Ulrich et al., 2009), suggesting a role for the contractile force in regulating the cellular response to stiff matrices such as elevated proliferation. Hence, although the exact molecular mechanisms have not yet been fully elucidated across a diverse range of cancers, emerging evidence advocates that mechanics have a fundamental role in human cancer.
Furthermore, the links between Rho- and myosin-mediated contractility in regulating both cancer cell and stem cell behavior, and the potential role for adult stem or progenitor cells in human cancer raises additional questions regarding the interplay between mechanics and tumor initiation. As some human cancers, or a subset of cancers in a particular organ, have been proposed to result from aberrant adult tissue progenitor cells (Stingl and Caldas, 2007; Visvader and Lindeman, 2008), it is reasonable to assume that the mechanical properties of the microenvironment influences progenitor cell differentiation, as was shown for mesenchymal stem cells (Engler et al., 2006). Evidence that abnormal ECM stiffness disrupts differentiation and contributes to transformation is provided by the observation that altering stiffness and fibronectin levels in the ECM can control both normal acinar differentiation as well as over-proliferation of breast cells (Williams et al., 2008). Loss of normal intestinal epithelial differentiation, and a switch to enhanced proliferation and tumor formation is also observed in mice that lack decorin (Bi et al., 2008), a proteoglycan that has a role in collagen fibrillogenesis and organization. It makes sense that a substantial increase in the turnover rate of progenitor cells due to elevated proliferation induced by aberrant matrix stiffness makes these cells more susceptible to events that promote transformation. Thus, although the exact mechanisms by which adult progenitor cells are regulated are currently not known, the mechanical properties of the microenvironment are likely to have a significant influence over their behavior and, thus, offer a unifying principle in understanding cancers in which progenitor cells have a role.
Mechanical signals and breast cancer risk – a case for mechanotransduction in breast cancer risk associated with high breast tissue density
High mammographic density of breast tissue has been correlated with a greater than fourfold increase in the relative risk for developing breast cancer, making high density of breast tissue one of the most significant independent risk factors for developing the disease (Boyd et al., 1998; McCormack and dos Santos Silva, 2006). In fact, high mammographic density throughout the breast may account for up to 30% of breast cancers, whereas mutations in BRCA1 or BRCA2 – although conferring a greater relative risk – account for only 5% of total breast cancers [see Boyd et al. (Boyd et al., 2005) and references therein]. Furthermore, a recent study revealed that ductal carcinoma in situ occurs overwhelmingly in dense regions of the breast (Ursin et al., 2005), suggesting that local densities in breast tissue increase cancer risk.
Importantly, regions of increased breast density have substantially increased deposition of fibrillar collagen and collagen-associated proteoglycans such as decorin (Alowami et al., 2003; Guo et al., 2001; Li et al., 2005). Until recently, the link between mammographically dense breast tissue and the composition of breast tissue had only been correlative, as there was no direct evidence for a causative link. Therefore, we set out to test whether increased stromal collagen is sufficient to promote mammary carcinoma. Using transgenic mouse models with increased stromal collagen, we indeed found a causal link between breast tissue density and breast carcinoma, because increased collagen promotes tumor formation, growth and progression (Provenzano et al., 2008a). It is interesting that one of the primary factors that promotes mammary epithelial cell proliferation and invasion in collagen dense 3D microenvironments is the high stiffness of these collagen matrices (Provenzano et al., 2009). The stiff ECM resists cellular contraction, resulting in elevated Rho-dependent intracellular contractile force, integrin clustering and FAK-dependent proliferation mediated by ERKs. These observations suggest that a chronically stiff microenvironment results in the increased (pre-tumor) epithelial content that is found in patients with high density of breast tissue (Guo et al., 2001; Li et al., 2005). Moreover, abnormally elevated cell growth and cell turnover that occurs in a stiff microenvironment might be subjected to mutagenic damage arising from chronic inflammatory disease (Martin and Boyd, 2008), leading to transformation. Furthermore, the emerging role for mammary progenitor cells that reside in the basal compartment of the mammary gland in breast cancer (Stingl and Caldas, 2007) raises the interesting possibility that a collagen-dense (stiff) stroma leads to hyperproliferation of progenitor cells in women with high density of breast tissue, ultimately contributing to the transition to carcinoma.
In addition to a direct effect on mammary epithelial cell growth, the stiff collagen-dense stroma in women with high density of breast tissue is likely to also promote an activated stromal cell population (commonly referred to as ‘reactive’ stroma), i.e. upregulation of growth factor, cytokine and/or excessive ECM secretion by the stromal cell population that is often associated with a pathological condition. Consistent with this hypothesis, fibroblast proliferation is amplified in 3D environments that resist the contractile force of the cell (Fringer and Grinnell, 2001; Rosenfeldt and Grinnell, 2000), and higher numbers of fibroblasts are present in high-density breast tissue (Alowami et al., 2003; Guo et al., 2001). Mechanical signals may promote the secretion of mitogenic factors by activating the fibroblasts that contribute to abnormal epithelial behavior in collagen-dense tissue because it is known that stromal fibroblasts can regulate the epithelium, in part through secreting growth factors and cytokines (Allinen et al., 2004; Bavik et al., 2006; Orimo et al., 2005). Furthermore, chronic inflammation is known to promote tumor progression (Condeelis and Pollard, 2006) and fibroblast activation might help recruit immune components that can promote activated stroma. Hence, it is likely that the stiff environment associated with high density of breast tissue activates a series of related mechanotransduction cascades in both epithelial and stromal cells throughout the breast, which result in chemical changes in the breast and in paracrine signaling that drives the epithelium towards transformation.
Conclusions
Here we have discussed the mechanisms by which mechanical signals, particularly those related to 3D matrix mechanical properties, regulate cellular responses such as cell proliferation. Cell proliferation is subject to many levels of control, but it is becoming clear that mechanical signaling through the cytoskeleton linkage between FAs and regulators of cellular contractility contribute to the regulation of cell proliferation. In addition, mechanical signals that are associated with cell and matrix changes during cancer formation and progression have a fundamental role in tumor growth and invasion. Moreover, a stiff extracellular matrix will contribute to the activation and proliferation of tumor-associated stromal cells. All of these factors might support carcinoma growth and progression in general, and in dense breast tissue in particular. Thus, although our understanding of the physical mechanisms of force transmission in cells and the underlying molecular mechanisms of mechanotransduction remains incomplete, it is clear that these processes are fundamental regulators of cell behavior. As such, carefully executed studies to determine the mechanical properties of the cell microenvironment in vivo and the use of 3D culture systems that closely mimic these properties will shed substantial light on the mechanisms by which mechanical signals regulate cell behavior.
Acknowledgments
We thank the members of the Keely and LOCI Laboratories for helpful discussions and comments on this article. This work was supported by grants from the National Institutes of Health (NIH): CA152249 to P.P.P. and CA142833 and CA114462 to P.J.K. Deposited in PMC for release after 12 months.
References
- Aijaz S., D'Atri F., Citi S., Balda M. S., Matter K. (2005). Binding of GEF-H1 to the tight junction-associated adaptor cingulin results in inhibition of Rho signaling and G1/S phase transition. Dev. Cell 8, 777-786 [DOI] [PubMed] [Google Scholar]
- Alcaraz J., Xu R., Mori H., Nelson C. M., Mroue R., Spencer V. A., Brownfield D., Radisky D. C., Bustamante C., Bissell M. J. (2008). Laminin and biomimetic extracellular elasticity enhance functional differentiation in mammary epithelia. EMBO J. 27, 2829-2838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allinen M., Beroukhim R., Cai L., Brennan C., Lahti-Domenici J., Huang H., Porter D., Hu M., Chin L., Richardson A., et al. (2004). Molecular characterization of the tumor microenvironment in breast cancer. Cancer Cell 6, 17-32 [DOI] [PubMed] [Google Scholar]
- Alowami S., Troup S., Al-Haddad S., Kirkpatrick I., Watson P. H. (2003). Mammographic density is related to stroma and stromal proteoglycan expression. Breast Cancer Res. 5, R129-R135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amatangelo M. D., Bassi D. E., Klein-Szanto A. J., Cukierman E. (2005). Stroma-derived three-dimensional matrices are necessary and sufficient to promote desmoplastic differentiation of normal fibroblasts. Am. J. Pathol. 167, 475-488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aplin A. E., Juliano R. L. (1999). Integrin and cytoskeletal regulation of growth factor signaling to the MAP kinase pathway. J. Cell Sci. 112, 695-706 [DOI] [PubMed] [Google Scholar]
- Arthur W. T., Burridge K. (2001). RhoA inactivation by p190RhoGAP regulates cell spreading and migration by promoting membrane protrusion and polarity. Mol. Biol. Cell 12, 2711-2720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arthur W. T., Petch L. A., Burridge K. (2000). Integrin engagement suppresses RhoA activity via a c-Src-dependent mechanism. Curr. Biol. 10, 719-722 [DOI] [PubMed] [Google Scholar]
- Assoian R. K., Klein E. A. (2008). Growth control by intracellular tension and extracellular stiffness. Trends Cell Biol. 18, 347-352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron V., Calleja V., Ferrari P., Alengrin F., Van Obberghen E. (1998). p125Fak focal adhesion kinase is a substrate for the insulin and insulin-like growth factor-I tyrosine kinase receptors. J. Biol. Chem. 273, 7162-7168 [DOI] [PubMed] [Google Scholar]
- Baselga J., Swain S. M. (2009). Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat. Rev. Cancer 9, 463-475 [DOI] [PubMed] [Google Scholar]
- Basson M. D., Yu C. F., Herden-Kirchoff O., Ellermeier M., Sanders M. A., Merrell R. C., Sumpio B. E. (2000). Effects of increased ambient pressure on colon cancer cell adhesion. J. Cell. Biochem. 78, 47-61 [DOI] [PubMed] [Google Scholar]
- Bavik C., Coleman I., Dean J. P., Knudsen B., Plymate S., Nelson P. S. (2006). The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Res. 66, 794-802 [DOI] [PubMed] [Google Scholar]
- Benlimame N., He Q., Jie S., Xiao D., Xu Y. J., Loignon M., Schlaepfer D. D., Alaoui-Jamali M. A. (2005). FAK signaling is critical for ErbB-2/ErbB-3 receptor cooperation for oncogenic transformation and invasion. J. Cell Biol. 171, 505-516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bi X., Tong C., Dockendorff A., Bancroft L., Gallagher L., Guzman G., Iozzo R. V., Augenlicht L. H., Yang W. (2008). Genetic deficiency of decorin causes intestinal tumor formation through disruption of intestinal cell maturation. Carcinogenesis 29, 1435-1440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birukova A. A., Fu P., Xing J., Yakubov B., Cokic I., Birukov K. G. (2010). Mechanotransduction by GEF-H1 as a novel mechanism of ventilator induced vascular endothelial permeability. Am. J. Physiol. Lung Cell. Mol. Physiol. 298, 837-848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhadriraju K., Yang M., Alom Ruiz S., Pirone D., Tan J., Chen C. S. (2007). Activation of ROCK by RhoA is regulated by cell adhesion, shape, and cytoskeletal tension. Exp. Cell Res. 313, 3616-3623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd N. F., Lockwood G. A., Byng J. W., Tritchler D. L., Yaffe M. J. (1998). Mammographic densities and breast cancer risk. Cancer Epidemiol. Biomarkers Prev. 7, 1133-1144 [PubMed] [Google Scholar]
- Boyd N. F., Rommens J. M., Vogt K., Lee V., Hopper J. L., Yaffe M. J., Paterson A. D. (2005). Mammographic breast density as an intermediate phenotype for breast cancer. Lancet Oncol. 6, 798-808 [DOI] [PubMed] [Google Scholar]
- Brandt D. T., Baarlink C., Kitzing T. M., Kremmer E., Ivaska J., Nollau P., Grosse R. (2009). SCAI acts as a suppressor of cancer cell invasion through the transcriptional control of beta1-integrin. Nat. Cell Biol. 11, 557-568 [DOI] [PubMed] [Google Scholar]
- Burkard M. E., Randall C. L., Larochelle S., Zhang C., Shokat K. M., Fisher R. P., Jallepalli P. V. (2007). Chemical genetics reveals the requirement for Polo-like kinase 1 activity in positioning RhoA and triggering cytokinesis in human cells. Proc. Natl. Acad. Sci. 104, 4383-4388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burridge K., Fath K., Kelly T., Nuckolls G., Turner C. (1988). Focal adhesions: transmembrane junctions between the extracellular matrix and the cytoskeleton. Annu. Rev. Cell Biol. 4, 487-525 [DOI] [PubMed] [Google Scholar]
- Casamassima A., Rozengurt E. (1998). Insulin-like growth factor I stimulates tyrosine phosphorylation of p130(Cas), focal adhesion kinase, and paxillin. Role of phosphatidylinositol 3′-kinase and formation of a p130(Cas).Crk complex. J. Biol. Chem. 273, 26149-26156 [DOI] [PubMed] [Google Scholar]
- Chen C. S., Mrksich M., Huang S., Whitesides G. M., Ingber D. E. (1997). Geometric control of cell life and death. Science 276, 1425-1428 [DOI] [PubMed] [Google Scholar]
- Chen C. S., Tan J., Tien J. (2004). Mechanotransduction at cell-matrix and cell-cell contacts. Annu. Rev. Biomed. Eng. 6, 275-302 [DOI] [PubMed] [Google Scholar]
- Choquet D., Felsenfeld D. P., Sheetz M. P. (1997). Extracellular matrix rigidity causes strengthening of integrin-cytoskeleton linkages. Cell 88, 39-48 [DOI] [PubMed] [Google Scholar]
- Chrzanowska-Wodnicka M., Burridge K. (1996). Rho-stimulated contractility drives the formation of stress fibers and focal adhesions. J. Cell Biol. 133, 1403-1415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Citi S., Paschoud S., Pulimeno P., Timolati F., De Robertis F., Jond L., Guillemot L. (2009). The tight junction protein cingulin regulates gene expression and RhoA signaling. Ann. N. Y. Acad. Sci. 1165, 88-98 [DOI] [PubMed] [Google Scholar]
- Condeelis J., Pollard J. W. (2006). Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124, 263-266 [DOI] [PubMed] [Google Scholar]
- Cukierman E., Bassi D. E. (2010). Physico-mechanical aspects of extracellular matrix influences on tumorigenic behaviors. Semin. Cancer Biol. 20, 139-145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cukierman E., Pankov R., Stevens D. R., Yamada K. M. (2001). Taking cell-matrix adhesions to the third dimension. Science 294, 1708-1712 [DOI] [PubMed] [Google Scholar]
- Discher D. E., Janmey P., Wang Y. L. (2005). Tissue cells feel and respond to the stiffness of their substrate. Science 310, 1139-1143 [DOI] [PubMed] [Google Scholar]
- Engler A. J., Sen S., Sweeney H. L., Discher D. E. (2006). Matrix elasticity directs stem cell lineage specification. Cell 126, 677-689 [DOI] [PubMed] [Google Scholar]
- Engler A. J., Carag-Krieger C., Johnson C. P., Raab M., Tang H. Y., Speicher D. W., Sanger J. W., Sanger J. M., Discher D. E. (2008). Embryonic cardiomyocytes beat best on a matrix with heart-like elasticity: scar-like rigidity inhibits beating. J. Cell Sci. 121, 3794-3802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faried A., Faried L. S., Usman N., Kato H., Kuwano H. (2007). Clinical and prognostic significance of RhoA and RhoC gene expression in esophageal squamous cell carcinoma. Ann. Surg. Oncol. 14, 3593-3601 [DOI] [PubMed] [Google Scholar]
- Fischer R. S., Gardel M., Ma X., Adelstein R. S., Waterman C. M. (2009). Local cortical tension by myosin II guides 3D endothelial cell branching. Curr. Biol. 19, 260-265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fluck M., Carson J. A., Gordon S. E., Ziemiecki A., Booth F. W. (1999). Focal adhesion proteins FAK and paxillin increase in hypertrophied skeletal muscle. Am. J. Physiol. 277, C152-C162 [DOI] [PubMed] [Google Scholar]
- Folkman J., Moscona A. (1978). Role of cell shape in growth control. Nature 273, 345-349 [DOI] [PubMed] [Google Scholar]
- Fraley S. I., Feng Y., Krishnamurthy R., Kim D. H., Celedon A., Longmore G. D., Wirtz D. (2010). A distinctive role for focal adhesion proteins in three-dimensional cell motility. Nat. Cell Biol. 12, 598-604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fringer J., Grinnell F. (2001). Fibroblast quiescence in floating or released collagen matrices: contribution of the ERK signaling pathway and actin cytoskeletal organization. J. Biol. Chem. 276, 31047-31052 [DOI] [PubMed] [Google Scholar]
- Fringer J., Grinnell F. (2003). Fibroblast quiescence in floating collagen matrices-Decrease in serum activation of MEK and RAF but not Ras. J. Biol. Chem. 278, 20612-20617 [DOI] [PubMed] [Google Scholar]
- Galbraith C. G., Yamada K. M., Sheetz M. P. (2002). The relationship between force and focal complex development. J. Cell Biol. 159, 695-705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardel M. L., Nakamura F., Hartwig J., Crocker J. C., Stossel T. P., Weitz D. A. (2006). Stress-dependent elasticity of composite actin networks as a model for cell behavior. Phys. Rev. Lett. 96, 088102 [DOI] [PubMed] [Google Scholar]
- Gardel M. L., Sabass B., Ji L., Danuser G., Schwarz U. S., Waterman C. M. (2008). Traction stress in focal adhesions correlates biphasically with actin retrograde flow speed. J. Cell Biol. 183, 999-1005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehler S., Baldassarre M., Lad Y., Leight J. L., Wozniak M. A., Riching K. M., Eliceiri K. W., Weaver V. M., Calderwood D. A., Keely P. J. (2009). Filamin A-beta1 integrin complex tunes epithelial cell response to matrix tension. Mol. Biol. Cell 20, 3224-3238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geiger B., Bershadsky A., Pankov R., Yamada K. M. (2001). Transmembrane crosstalk between the extracellular matrix-cytoskeleton crosstalk. Nat. Rev. Mol. Cell Biol. 2, 793-805 [DOI] [PubMed] [Google Scholar]
- Gineitis D., Treisman R. (2001). Differential usage of signal transduction pathways defines two types of serum response factor target gene. J. Biol. Chem. 276, 24531-24539 [DOI] [PubMed] [Google Scholar]
- Glotzer M. (2005). The molecular requirements for cytokinesis. Science 307, 1735-1739 [DOI] [PubMed] [Google Scholar]
- Golubovskaya V. M., Finch R., Cance W. G. (2005). Direct interaction of the N-terminal domain of focal adhesion kinase with the N-terminal transactivation domain of p53. J. Biol. Chem. 280, 25008-25021 [DOI] [PubMed] [Google Scholar]
- Gopinath S. D., Narumiya S., Dhawan J. (2007). The RhoA effector mDiaphanous regulates MyoD expression and cell cycle progression via SRF-dependent and SRF-independent pathways. J. Cell Sci. 120, 3086-3098 [DOI] [PubMed] [Google Scholar]
- Gordon S. E., Fluck M., Booth F. W. (2001). Selected Contribution: Skeletal muscle focal adhesion kinase, paxillin, and serum response factor are loading dependent. J. Appl. Physiol. 90, 1174-83; discussion 1165 [DOI] [PubMed] [Google Scholar]
- Grinnell F. (2003). Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol. 13, 264-269 [DOI] [PubMed] [Google Scholar]
- Grinnell F., Ho C. H., Tamariz E., Lee D. J., Skuta G. (2003). Dendritic fibroblasts in three-dimensional collagen matrices. Mol. Biol. Cell 14, 384-395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y. P., Martin L. J., Hanna W., Banerjee D., Miller N., Fishell E., Khokha R., Boyd N. F. (2001). Growth factors and stromal matrix proteins associated with mammographic densities. Cancer Epidemiol. Biomarkers Prev. 10, 243-248 [PubMed] [Google Scholar]
- Hadjipanayi E., Mudera V., Brown R. A. (2009). Guiding cell migration in 3D: a collagen matrix with graded directional stiffness. Cell Motil. Cytoskeleton 66, 121-128 [DOI] [PubMed] [Google Scholar]
- Hahn C., Schwartz M. A. (2009). Mechanotransduction in vascular physiology and atherogenesis. Nat. Rev. Mol. Cell Biol. 10, 53-62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halka A. T., Turner N. J., Carter A., Ghosh J., Murphy M. O., Kirton J. P., Kielty C. M., Walker M. G. (2008). The effects of stretch on vascular smooth muscle cell phenotype in vitro. Cardiovasc. Pathol. 17, 98-102 [DOI] [PubMed] [Google Scholar]
- He H., Yim M., Liu K. H., Cody S. C., Shulkes A., Baldwin G. S. (2008). Involvement of G proteins of the Rho family in the regulation of Bcl-2-like protein expression and caspase 3 activation by Gastrins. Cell. Signal. 20, 83-93 [DOI] [PubMed] [Google Scholar]
- Helm C. L., Zisch A., Swartz M. A. (2007). Engineered blood and lymphatic capillaries in 3-D VEGF-fibrin-collagen matrices with interstitial flow. Biotechnol. Bioeng. 96, 167-176 [DOI] [PubMed] [Google Scholar]
- Hill C. S., Wynne J., Treisman R. (1995). The Rho family GTPases RhoA, Rac1, and CDC42Hs regulate transcriptional activation by SRF. Cell 81, 1159-1170 [DOI] [PubMed] [Google Scholar]
- Huang S., Chen C. S., Ingber D. E. (1998). Control of cyclin D1, p27(Kip1), and cell cycle progression in human capillary endothelial cells by cell shape and cytoskeletal tension. Mol. Biol. Cell 9, 3179-3193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingber D. E. (2006). Cellular mechanotransduction: putting all the pieces together again. FASEB J. 20, 811-827 [DOI] [PubMed] [Google Scholar]
- Ishibe S., Joly D., Zhu X., Cantley L. G. (2003). Phosphorylation-dependent paxillin-ERK association mediates hepatocyte growth factor-stimulated epithelial morphogenesis. Mol. Cell 12, 1275-1285 [DOI] [PubMed] [Google Scholar]
- Ishibe S., Joly D., Liu Z. X., Cantley L. G. (2004). Paxillin serves as an ERK-regulated scaffold for coordinating FAK and Rac activation in epithelial morphogenesis. Mol. Cell 16, 257-267 [DOI] [PubMed] [Google Scholar]
- Janmey P. A., McCulloch C. A. (2007). Cell mechanics: integrating cell responses to mechanical stimuli. Annu. Rev. Biomed. Eng. 9, 1-34 [DOI] [PubMed] [Google Scholar]
- Junttila M. R., Evan G. I. (2009). p53-a Jack of all trades but master of none. Nat. Rev. Cancer 9, 821-829 [DOI] [PubMed] [Google Scholar]
- Klein E. A., Yin L., Kothapalli D., Castagnino P., Byfield F. J., Xu T., Levental I., Hawthorne E., Janmey P. A., Assoian R. K. (2009). Cell-cycle control by physiological matrix elasticity and in vivo tissue stiffening. Curr. Biol. 19, 1511-1518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koyama H., Raines E. W., Bornfeldt K. E., Roberts J. M., Ross R. (1996). Fibrillar collagen inhibits arterial smooth muscle proliferation through regulation of Cdk2 inhibitors. Cell 87, 1069-1078 [DOI] [PubMed] [Google Scholar]
- Kumar A., Murphy R., Robinson P., Wei L., Boriek A. M. (2004). Cyclic mechanical strain inhibits skeletal myogenesis through activation of focal adhesion kinase, Rac-1 GTPase, and NF-kappaB transcription factor. FASEB J. 18, 1524-1535 [DOI] [PubMed] [Google Scholar]
- Kuwahara K., Barrientos T., Pipes G. C., Li S., Olson E. N. (2005). Muscle-specific signaling mechanism that links actin dynamics to serum response factor. Mol. Cell. Biol. 25, 3173-3181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H. S., Millward-Sadler S. J., Wright M. O., Nuki G., Salter D. M. (2000). Integrin and mechanosensitive ion channel-dependent tyrosine phosphorylation of focal adhesion proteins and beta-catenin in human articular chondrocytes after mechanical stimulation. J. Bone Miner. Res. 15, 1501-1509 [DOI] [PubMed] [Google Scholar]
- Leucht P., Kim J. B., Currey J. A., Brunski J., Helms J. A. (2007). FAK-Mediated mechanotransduction in skeletal regeneration. PLoS One 2, e390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F., Guo W. Y., Li W. J., Zhang D. X., Lv A. L., Luan R. H., Liu B., Wang H. C. (2009). Cyclic stretch upregulates SDF-1alpha/CXCR4 axis in human saphenous vein smooth muscle cells. Biochem. Biophys. Res. Commun. 386, 247-251 [DOI] [PubMed] [Google Scholar]
- Li T., Sun L., Miller N., Nicklee T., Woo J., Hulse-Smith L., Tsao M. S., Khokha R., Martin L., Boyd N. (2005). The association of measured breast tissue characteristics with mammographic density and other risk factors for breast cancer. Cancer Epidemiol. Biomarkers Prev. 14, 343-349 [DOI] [PubMed] [Google Scholar]
- Lim S. T., Chen X. L., Lim Y., Hanson D. A., Vo T. T., Howerton K., Larocque N., Fisher S. J., Schlaepfer D. D., Ilic D. (2008). Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation. Mol. Cell 29, 9-22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Gardel M. L., Kroy K., Frey E., Hoffman B. D., Crocker J. C., Bausch A. R., Weitz D. A. (2006). Microrheology probes length scale dependent rheology. Phys. Rev. Lett. 96, 118104 [DOI] [PubMed] [Google Scholar]
- Liu Z. X., Yu C. F., Nickel C., Thomas S., Cantley L. G. (2002). Hepatocyte growth factor induces ERK-dependent paxillin phosphorylation and regulates paxillin-focal adhesion kinase association. J. Biol. Chem. 277, 10452-10458 [DOI] [PubMed] [Google Scholar]
- Lo C. M., Wang H. B., Dembo M., Wang L. Y. (2000). Cell movement is guided by the rigidity of the substrate. Biophys. J. 79, 144-152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockman K., Hinson J. S., Medlin M. D., Morris D., Taylor J. M., Mack C. P. (2004). Sphingosine 1-phosphate stimulates smooth muscle cell differentiation and proliferation by activating separate serum response factor co-factors. J. Biol. Chem. 279, 42422-42430 [DOI] [PubMed] [Google Scholar]
- Luan Y., Lieleg O., Wagner B., Bausch A. R. (2008). Micro- and macrorheological properties of isotropically cross-linked actin networks. Biophys. J. 94, 688-693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Z., Liu Z., Myers D. P., Terada L. S. (2008). Mechanotransduction and anoikis: death and the homeless cell. Cell Cycle 7, 2462-2465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mammoto A., Connor K. M., Mammoto T., Yung C. W., Huh D., Aderman C. M., Mostoslavsky G., Smith L. E., Ingber D. E. (2009). A mechanosensitive transcriptional mechanism that controls angiogenesis. Nature 457, 1103-1108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin L. J., Boyd N. F. (2008). Mammographic density. Potential mechanisms of breast cancer risk associated with mammographic density: hypotheses based on epidemiological evidence. Breast Cancer Res. 10, 201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBeath R., Pirone D. M., Nelson C. M., Bhadriraju K., Chen C. S. (2004). Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 6, 483-495 [DOI] [PubMed] [Google Scholar]
- McCormack V. A., dos Santos Silva I. (2006). Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol. Biomarkers Prev. 15, 1159-1169 [DOI] [PubMed] [Google Scholar]
- Medjkane S., Perez-Sanchez C., Gaggioli C., Sahai E., Treisman R. (2009). Myocardin-related transcription factors and SRF are required for cytoskeletal dynamics and experimental metastasis. Nat. Cell Biol. 11, 257-268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minden A., Lin A., Claret F. X., Abo A., Karin M. (1995). Selective activation of the JNK signaling cascade and c-Jun transcriptional activity by the small GTPases Rac and Cdc42Hs. Cell 81, 1147-1157 [DOI] [PubMed] [Google Scholar]
- Mitra S. K., Hanson D. A., Schlaepfer D. D. (2005). Focal adhesion kinase: in command and control of cell motility. Nat. Rev. Mol. Cell Biol. 6, 56-68 [DOI] [PubMed] [Google Scholar]
- Moalli M. R., Wang S., Caldwell N. J., Patil P. V., Maynard C. R. (2001). Mechanical stimulation induces pp125(FAK) and pp60(src) activity in an in vivo model of trabecular bone formation. J. Appl. Physiol. 91, 912-918 [DOI] [PubMed] [Google Scholar]
- Molina T., Kabsch K., Alonso A., Kohl A., Komposch G., Tomakidi P. (2001). Topographic changes of focal adhesion components and modulation of p125FAK activation in stretched human periodontal ligament fibroblasts. J. Dent. Res. 80, 1984-1989 [DOI] [PubMed] [Google Scholar]
- Moore K. A., Polte T., Huang S., Shi B., Alsberg E., Sunday M. E., Ingber D. E. (2005). Control of basement membrane remodeling and epithelial branching morphogenesis in embryonic lung by Rho and cytoskeletal tension. Dev. Dyn. 232, 268-281 [DOI] [PubMed] [Google Scholar]
- Nakayama K., Obara K., Tanabe Y., Saito M., Ishikawa T., Nishizawa S. (2003). Interactive role of tyrosine kinase, protein kinase C, and Rho/Rho kinase systems in the mechanotransduction of vascular smooth muscles. Biorheology 40, 307-314 [PubMed] [Google Scholar]
- Neisch A. L., Speck O., Stronach B., Fehon R. G. (2010). Rho1 regulates apoptosis via activation of the JNK signaling pathway at the plasma membrane. J. Cell Biol. 189, 311-323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng C. P., Hinz B., Swartz M. A. (2005). Interstitial fluid flow induces myofibroblast differentiation and collagen alignment in vitro. J. Cell Sci. 118, 4731-4739 [DOI] [PubMed] [Google Scholar]
- Nie M., Aijaz S., Leefa Chong San I. V., Balda M. S., Matter K. (2009). The Y-box factor ZONAB/DbpA associates with GEF-H1/Lfc and mediates Rho-stimulated transcription. EMBO Rep. 10, 1125-1131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olson E. N., Nordheim A. (2010). Linking actin dynamics and gene transcription to drive cellular motile functions. Nat. Rev. Mol. Cell Biol. 11, 353-365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orimo A., Gupta P. B., Sgroi D. C., Arenzana-Seisdedos F., Delaunay T., Naeem R., Carey V. J., Richardson A. L., Weinberg R. A. (2005). Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335-348 [DOI] [PubMed] [Google Scholar]
- Parsons S. J., Parsons J. T. (2004). Src family kinases, key regulators of signal transduction. Oncogene 23, 7906-7909 [DOI] [PubMed] [Google Scholar]
- Paszek M. J., Zahir N., Johnson K. R., Lakins J. N., Rozenberg G. I., Gefen A., Reinhart-King C. A., Margulies S. S., Dembo M., Boettiger D., et al. (2005). Tensional homeostasis and the malignant phenotype. Cancer Cell 8, 241-254 [DOI] [PubMed] [Google Scholar]
- Pelham R. J., Jr, Wang Y. (1997). Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc. Natl. Acad. Sci. USA 94, 13661-13665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petronczki M., Glotzer M., Kraut N., Peters J.-M. (2007). Polo-like kinase 1 triggers the initiation of cytokinesis in human cells by promoting recruitment of the RhoGEF Ect2 to the central spindle. Dev. Cell 12, 713-725 [DOI] [PubMed] [Google Scholar]
- Philips A., Roux P., Coulon V., Bellanger J. M., Vie A., Vignais M. L., Blanchard J. M. (2000). Differential effect of Rac and Cdc42 on p38 kinase activity and cell cycle progression of nonadherent primary mouse fibroblasts. J. Biol. Chem. 275, 5911-5917 [DOI] [PubMed] [Google Scholar]
- Pirone D. M., Liu W. F., Ruiz S. A., Gao L., Raghavan S., Lemmon C. A., Romer L. H., Chen C. S. (2006). An inhibitory role for FAK in regulating proliferation: a link between limited adhesion and RhoA-ROCK signaling. J. Cell Biol. 174, 277-288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Playford M. P., Schaller M. D. (2004). The interplay between Src and integrins in normal and tumor biology. Oncogene 23, 7928-7946 [DOI] [PubMed] [Google Scholar]
- Pollak M. (2008). Insulin and insulin-like growth factor signalling in neoplasia. Nat. Rev. Cancer 8, 915-928 [DOI] [PubMed] [Google Scholar]
- Provenzano P. P., Inman D. R., Eliceiri K. W., Knittel J. G., Yan L., Rueden C. T., White J. G., Keely P. J. (2008a). Collagen density promotes mammary tumor initiation and progression. BMC Med. 6, 11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Provenzano P. P., Inman D. R., Eliceiri K. W., Trier S. M., Keely P. J. (2008b). Contact guidance mediated three-dimensional cell migration is regulated by Rho/ROCK-dependent matrix reorganization. Biophys. J. 95, 5374-5384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Provenzano P. P., Inman D. R., Eliceiri K. W., Keely P. J. (2009). Matrix density-induced mechanoregulation of breast cell phenotype, signaling and gene expression through a FAK-ERK linkage. Oncogene 28, 4326-4343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruitt K., Der C. J. (2001). Ras and Rho regulation of the cell cycle and oncogenesis. Cancer Lett. 171, 1-10 [DOI] [PubMed] [Google Scholar]
- Qi Y. X., Qu M. J., Yan Z. Q., Zhao D., Jiang X. H., Shen B. R., Jiang Z. L. (2010). Cyclic strain modulates migration and proliferation of vascular smooth muscle cells via Rho-GDIalpha, Rac1, and p38 pathway. J. Cell. Biochem. 109, 906-914 [DOI] [PubMed] [Google Scholar]
- Ridley A. J., Hall A. (1992). The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 70, 389-399 [DOI] [PubMed] [Google Scholar]
- Robling A. G., Castillo A. B., Turner C. H. (2006). Biomechanical and molecular regulation of bone remodeling. Annu. Rev. Biomed. Eng. 8, 455-498 [DOI] [PubMed] [Google Scholar]
- Rosenfeldt H., Grinnell F. (2000). Fibroblast quiescence and the disruption of ERK signaling in mechanically unloaded collagen matrices. J. Biol. Chem. 275, 3088-3092 [DOI] [PubMed] [Google Scholar]
- Saez A., Buguin A., Silberzan P., Ladoux B. (2005). Is the mechanical activity of epithelial cells controlled by deformations or forces? Biophys. J. 89, L52-L54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahai E., Marshall C. J. (2002). ROCK and Dia have opposing effects on adherens junctions downstream of Rho. Nat. Cell Biol. 4, 408-415 [DOI] [PubMed] [Google Scholar]
- Sawada Y., Tamada M., Dubin-Thaler B. J., Cherniavskaya O., Sakai R., Tanaka S., Sheetz M. P. (2006). Force sensing by mechanical extension of the Src family kinase substrate p130Cas. Cell 127, 1015-1026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaller M. D., Hildebrand J. D., Shannon J. D., Fox J. W., Vines R. R., Parsons J. T. (1994). Autophosphorylation of the focal adhesion kinase, pp125FAK, directs SH2-dependent binding of pp60src. Mol. Cell. Biol. 14, 1680-1688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer D. D., Hunter T. (1996). Evidence for in vivo phosphorylation of the Grb2 SH2-domain binding site on focal adhesion kinase by Src-family protein-tyrosine kinases. Mol. Cell. Biol. 16, 5623-5633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer D. D., Hanks S. K., Hunter T., van der Geer P. (1994). Integrin-mediated signal transduction linked to Ras pathway by GRB2 binding to focal adhesion kinase. Nature 372, 786-791 [DOI] [PubMed] [Google Scholar]
- Schlaepfer D. D., Jones K. C., Hunter T. (1998). Multiple Grb2-mediated integrin-stimulated signaling pathways to ERK2/mitogen-activated protein kinase: summation of both c-Src- and focal adhesion kinase-initiated tyrosine phosphorylation events. Mol. Cell. Biol. 18, 2571-2585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlessinger K., Hall A., Tolwinski N. (2009). Wnt signaling pathways meet Rho GTPases. Genes Dev. 23, 265-277 [DOI] [PubMed] [Google Scholar]
- Serebriiskii I., Castello-Cros R., Lamb A., Golemis E. A., Cukierman E. (2008). Fibroblast-derived 3D matrix differentially regulates the growth and drug-responsiveness of human cancer cells. Matrix Biol. 27, 573-585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieg D. J., Hauck C. R., Ilic D., Klingbeil C. K., Schaefer E., Damsky C. H., Schlaepfer D. D. (2000). FAK integrates growth-factor and integrin signals to promote cell migration. Nat. Cell Biol. 2, 249-256 [DOI] [PubMed] [Google Scholar]
- Sniadecki N. J., Anguelouch A., Yang M. T., Lamb C. M., Liu Z., Kirschner S. B., Liu Y., Reich D. H., Chen C. S. (2007). Magnetic microposts as an approach to apply forces to living cells. Proc. Natl. Acad. Sci. USA 104, 14553-14558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solon J., Levental I., Sengupta K., Georges P. C., Janmey P. A. (2007). Fibroblast adaptation and stiffness matching to soft elastic substrates. Biophys. J. 93, 4453-4461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterpetti P., Marucci L., Candelaresi C., Toksoz D., Alpini G., Ugili L., Baroni G. S., Macarri G., Benedetti A. (2006). Cell proliferation and drug resistance in hepatocellular carcinoma are modulated by Rho GTPase signals. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G624-G632 [DOI] [PubMed] [Google Scholar]
- Stingl J., Caldas C. (2007). Molecular heterogeneity of breast carcinomas and the cancer stem cell hypothesis. Nat. Rev. Cancer 7, 791-799 [DOI] [PubMed] [Google Scholar]
- Su L., Pertz O., Mikawa M., Hahn K., Parsons S. J. (2009). p190RhoGAP negatively regulates Rho activity at the cleavage furrow of mitotic cells. Exp. Cell Res. 315, 1347-1359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun H. W., Tong S. L., He J., Wang Q., Zou L., Ma S. J., Tan H. Y., Luo J. F., Wu H. X. (2007). RhoA and RhoC -siRNA inhibit the proliferation and invasiveness activity of human gastric carcinoma by Rho/PI3K/Akt pathway. World J. Gastroenterol. 13, 3517-3522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor W. R., Stark G. R. (2001). Regulation of the G2/M transition by p53. Oncogene 20, 1803-1815 [DOI] [PubMed] [Google Scholar]
- Tschumperlin D. J., Dai G., Maly I. V., Kikuchi T., Laiho L. H., McVittie A. K., Haley K. J., Lilly C. M., So P. T., Lauffenburger D. A., et al. (2004). Mechanotransduction through growth-factor shedding into the extracellular space. Nature 429, 83-86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng Y., An K. M., Esue O., Wirtz D. (2004). The bimodal role of filamin in controlling the architecture and mechanics of F-actin networks. J. Biol. Chem. 279, 1819-1826 [DOI] [PubMed] [Google Scholar]
- Ulrich T. A., de Juan Pardo E. M., Kumar S. (2009). The mechanical rigidity of the extracellular matrix regulates the structure, motility, and proliferation of glioma cells. Cancer Res. 69, 4167-4174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ursin G., Hovanessian-Larsen L., Parisky Y. R., Pike M. C., Wu A. H. (2005). Greatly increased occurrence of breast cancers in areas of mammographically dense tissue. Breast Cancer Res. 7, R605-R608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Visvader J. E., Lindeman G. J. (2008). Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat. Rev. Cancer 8, 755-768 [DOI] [PubMed] [Google Scholar]
- Vogel V., Sheetz M. (2006). Local force and geometry sensing regulate cell functions. Nat. Rev. Mol. Cell Biol. 7, 265-275 [DOI] [PubMed] [Google Scholar]
- Wang H. B., Dembo M., Wang Y. L. (2000). Substrate flexibility regulates growth and apoptosis of normal but not transformed cells. Am. J. Physiol. Cell Physiol. 279, C1345-C1350 [DOI] [PubMed] [Google Scholar]
- Wang H. B., Dembo M., Hanks S. K., Wang Y. (2001). Focal adhesion kinase is involved in mechanosensing during fibroblast migration. Proc. Natl. Acad. Sci. USA 98, 11295-11300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H. B., Liu X. P., Liang J., Yang K., Sui A. H., Liu Y. J. (2009). Expression of RhoA and RhoC in colorectal carcinoma and its relations with clinicopathological parameters. Clin. Chem. Lab. Med. 47, 811-817 [DOI] [PubMed] [Google Scholar]
- Wang J. G., Miyazu M., Matsushita E., Sokabe M., Naruse K. (2001). Uniaxial cyclic stretch induces focal adhesion kinase (FAK) tyrosine phosphorylation followed by mitogen-activated protein kinase (MAPK) activation. Biochem. Biophys. Res. Commun. 288, 356-361 [DOI] [PubMed] [Google Scholar]
- Wang J. G., Miyazu M., Xiang P., Li S. N., Sokabe M., Naruse K. (2005). Stretch-induced cell proliferation is mediated by FAK-MAPK pathway. Life Sci. 76, 2817-2825 [DOI] [PubMed] [Google Scholar]
- Wang N., Tytell J. D., Ingber D. E. (2009). Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat. Rev. Mol. Cell Biol. 10, 75-82 [DOI] [PubMed] [Google Scholar]
- Wang Z., Wang D. Z., Hockemeyer D., McAnally J., Nordheim A., Olson E. N. (2004). Myocardin and ternary complex factors compete for SRF to control smooth muscle gene expression. Nature 428, 185-189 [DOI] [PubMed] [Google Scholar]
- Webb D. J., Donais K., Whitmore L. A., Thomas S. M., Turner C. E., Parsons J. T., Horwitz A. F. (2004). FAK-Src signalling through paxillin, ERK and MLCK regulates adhesion disassembly. Nat. Cell Biol. 6, 154-161 [DOI] [PubMed] [Google Scholar]
- Welsh C. F., Roovers K., Villanueva J., Liu Y., Schwartz M. A., Assoian R. K. (2001). Timing of cyclin D1 expression within G1 phase is controlled by Rho. Nat. Cell Biol. 3, 950-957 [DOI] [PubMed] [Google Scholar]
- Whitehead I. P., Lambert Q. T., Glaven J. A., Abe K., Rossman K. L., Mahon G. M., Trzaskos J. M., Kay R., Campbell S. L., Der C. J. (1999). Dependence of Dbl and Dbs transformation on MEK and NF-kappaB activation. Mol. Cell. Biol. 19, 7759-7770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitfield M. L., George L. K., Grant G. D., Perou C. M. (2006). Common markers of proliferation. Nat. Rev. Cancer 6, 99-106 [DOI] [PubMed] [Google Scholar]
- Williams C. M., Engler A. J., Slone R. D., Galante L. L., Schwarzbauer J. E. (2008). Fibronectin expression modulates mammary epithelial cell proliferation during acinar differentiation. Cancer Res. 68, 3185-3192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf K., Mazo I., Leung H., Engelke K., von Andrian U. H., Deryugina E. I., Strongin A. Y., Brocker E. B., Friedl P. (2003). Compensation mechanism in tumor cell migration: mesenchymal-amoeboid transition after blocking of pericellular proteolysis. J. Cell Biol. 160, 267-277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wozniak M., Fausto A., Carron C. P., Meyer D. M., Hruska K. A. (2000). Mechanically strained cells of the osteoblast lineage organize their extracellular matrix through unique sites of alphavbeta3-integrin expression. J. Bone Miner. Res. 15, 1731-1745 [DOI] [PubMed] [Google Scholar]
- Wozniak M. A., Chen C. S. (2009). Mechanotransduction in development: a growing role for contractility. Nat. Rev. Mol. Cell Biol. 10, 34-43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wozniak M. A., Desai R., Solski P. A., Der C. J., Keely P. J. (2003). ROCK-generated contractility regulates breast epithelial cell differentiation in response to the physical properties of a three-dimensional collagen matrix. J. Cell Biol. 163, 583-595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing Z., Chen H. C., Nowlen J. K., Taylor S. J., Shalloway D., Guan J. L. (1994). Direct interaction of v-Src with the focal adhesion kinase mediated by the Src SH2 domain. Mol. Biol. Cell 5, 413-421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Tseng Y., Wirtz D. (2000). Strain hardening of actin filament networks. Regulation by the dynamic cross-linking protein alpha-actinin. J. Biol. Chem. 275, 35886-3592 [DOI] [PubMed] [Google Scholar]
- Zhang J., Li W., Sanders M. A., Sumpio B. E., Panja A., Basson M. D. (2003). Regulation of the intestinal epithelial response to cyclic strain by extracellular matrix proteins. FASEB J. 17, 926-928 [DOI] [PubMed] [Google Scholar]
- Zhang S., Tang Q., Xu F., Xue Y., Zhen Z., Deng Y., Liu M., Chen J., Liu S., Qiu M., et al. (2009). RhoA regulates G1-S progression of gastric cancer cells by modulation of multiple INK4 family tumor suppressors. Mol. Cancer Res. 7, 570-580 [DOI] [PubMed] [Google Scholar]